|
|
| | Bz-Tz-NHS Basic information |
| Product Name: | Bz-Tz-NHS | | Synonyms: | 5-Oxo-5-[[[4-(1,2,4,5-tetrazin-3-yl)phenyl]methyl]amino]pentanoic acid 2,5-dioxo-1-pyrrolidinyl ester;Benzylamino tetrazine N-hydroxysuccinimidyl ester;Benzylaminotetrazine-NHS;Bz-Tz-NHS;2,5-Dioxo-1-pyrrolidinyl 5-[4-(1,2,4,5-tetrazin-3-yl)benzylamino]-5-oxopentanoate;Tz-NHS;5-(4-(1,2,4,5-tetrazin-3-yl)benzylamino)-5-oxopentanoic acid succinimidyl ester;2,5-Dioxo-1-pyrrolidinyl 5-[4-(1,2,4,5-tetrazin-3-yl)benzylamino]-5-oxopentanoate >=90% | | CAS: | 1244040-64-9 | | MF: | C18H18N6O5 | | MW: | 398.37272 | | EINECS: | | | Product Categories: | | | Mol File: | 1244040-64-9.mol | 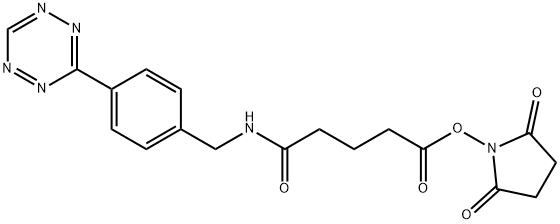 |
| | Bz-Tz-NHS Chemical Properties |
| density | 1.45±0.1 g/cm3(Predicted) | | storage temp. | -20°C | | solubility | DMSO : 100 mg/mL (251.02 mM; Need ultrasonic) | | pka | 14.57±0.46(Predicted) | | form | Solid | | color | Pink to red |
| Hazard Codes | Xn | | Risk Statements | 22 | | WGK Germany | 3 |
| | Bz-Tz-NHS Usage And Synthesis |
| Uses | Succinimidyl ester functionalized tetrazine for inverse electron demand Diels-Alder cycloaddition reactions. Succinimidyl ester will react with amines for small molecule, biomolecule or surface modification. Tetrazine will react with strained alkenes such as transcyclooctene, norbornene and cyclopropene to yield a stable covalent linkage. Tetrazines have proven useful in bioorthogonal reactions for many biological imaging and bioconjugation applications. |
| | Bz-Tz-NHS Preparation Products And Raw materials |
|