|
|
| | 2-hexyl-2-methyl-1,3-benzodioxole Basic information |
| Product Name: | 2-hexyl-2-methyl-1,3-benzodioxole | | Synonyms: | 2-hexyl-2-methyl-1,3-benzodioxole;2-Methyl-2-hexyl-1,3-benzodioxole;1,3-Benzodioxole, 2-hexyl-2-methyl-;Ai3-14236;Catechol, methyl hexyl ketal;Einecs 269-572-9;Pyrocatechin methyl hexyl ketal | | CAS: | 68298-48-6 | | MF: | C14H20O2 | | MW: | 220.31 | | EINECS: | 269-572-9 | | Product Categories: | | | Mol File: | 68298-48-6.mol | 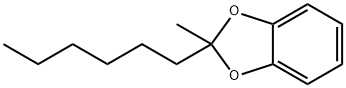 |
| | 2-hexyl-2-methyl-1,3-benzodioxole Chemical Properties |
| solubility | Practically insoluble in water, soluble in alcohol and oils | | color | Colorless oily liquid | | Odor | Sweet-floral, herbaceous, Jasmin-like odor of good tenacity | | LogP | 5.164 (est) | | EPA Substance Registry System | 1,3-Benzodioxole, 2-hexyl-2-methyl- (68298-48-6) |
| | 2-hexyl-2-methyl-1,3-benzodioxole Usage And Synthesis |
| General Description |
This ketal, one of many ketals developed from Catechol since the 1930s when a few of these new materials were marketed under trade names, has remained on the shelf in many perfume laboratories. Its odor offers an excellent extender to the odor of Amylcinnamic aldehyde in that it is more floral, less tenacious, less oily, but equally versatile and adjusts well to many floral fragrance types. If it were manufactured on a large scale, its price could undoubtedly be brought down within the attractive limits for use in everyday perfumery. It blends also well with Amylsalicylate, Hydroxycitronellal, Heliotropine, Linalool, etc and it is stable in mild alkali. It is produced from Catechol and Methyl hexyl ketone by condensation.
|
| | 2-hexyl-2-methyl-1,3-benzodioxole Preparation Products And Raw materials |
|