(DIMETHYLPHENYLSILYL)BORONIC ACID PINAC& manufacturers
|
| | (DIMETHYLPHENYLSILYL)BORONIC ACID PINAC& Basic information |
| Product Name: | (DIMETHYLPHENYLSILYL)BORONIC ACID PINAC& | | Synonyms: | (DIMETHYLPHENYLSILYL)BORONIC ACID PINAC&;(Dimethylphenylsilyl)boronic acid pinacol ester;B-(dimethylphenylsilyl)pinacolborane, 2-(Dimethylphenylsilyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane;(DiMethylphenylsilyl)boronic acid pinacol ester 95%;diMethylphenylsilyl boronic ester;1,3,2-Dioxaborolane,2-(dimethylphenylsilyl)-4,4,5,5-tetramethyl-;2-(PHENYLDIMETHYLSILYL)-4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLANE;2-(Dimethylphenylsilyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane > | | CAS: | 185990-03-8 | | MF: | C14H23BO2Si | | MW: | 262.23 | | EINECS: | | | Product Categories: | Organic boronic acid | | Mol File: | 185990-03-8.mol | 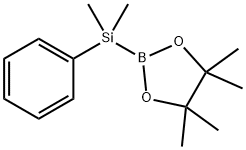 |
| | (DIMETHYLPHENYLSILYL)BORONIC ACID PINAC& Chemical Properties |
| Boiling point | 120°C/0.08mmHg(lit.) | | density | 0.962 g/mL at 25 °C | | refractive index | n20/D 1.4946 | | Fp | 110 °C | | storage temp. | Inert atmosphere,Room Temperature | | solubility | sol common organic solvents. | | form | clear liquid | | color | Colorless to Light yellow to Light orange | | Specific Gravity | 0.962 | | Hydrolytic Sensitivity | 7: reacts slowly with moisture/water |
| WGK Germany | 3 | | HS Code | 2934.99.4400 |
| | (DIMETHYLPHENYLSILYL)BORONIC ACID PINAC& Usage And Synthesis |
| Physical properties | bp 97–99? C/0.1 mmHg. | | Uses | 2-(Dimethylphenylsilyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is widely used as silaboration and silylation reagent for unsaturated organic com�pounds; a derivative having diethylamino group on the silicon
atom for use as a precursor for silylene generation. | | Uses | (Dimethylphenylsilyl)boronic acid pinacol ester (Suginome′s reagent) can be used as a reagent:
- For the selective addition of dimethylphenylsilanyl group to cyclic and acyclic unsaturated ketones, esters, acrylonitriles using a copper catalyst.
- In the synthesis of (Z)-4-boryl-1-silyl-2-alkene derivatives by stereoselective addition of silicon-boron bond to acyclic 1,3-dienes in presence of Ni catalyst.
- In the preparation of silyl-substituted butenoate and β-silyl-substituted acrylate derivatives from allenes and propiolate derivatives via hydrosilylation reactions using a copper catalyst.
- In the palladium-catalyzed asymmetric silaboration of allenes and alkanes to offered corresponding β-borylallylsilanes and 2-boryl-1-silylalkanes respectively.
| | Preparation | 2-(Dimethylphenylsilyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
(1) is prepared by reaction of dimethylphenylsilyllithium with
4,4,5,5-tetramethyl-1,3,2-dioxaborolane (pinacolborane, 2 equiv)
or 2-isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
(2 equiv). 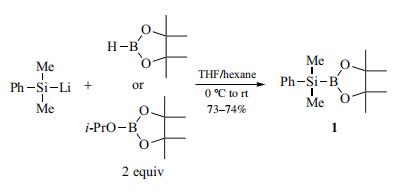
|
| | (DIMETHYLPHENYLSILYL)BORONIC ACID PINAC& Preparation Products And Raw materials |
| Raw materials | Boranediamine, 1-(dimethylphenylsilyl)-N,N,N',N'-tetraethyl- (9CI)-->Lithium, (dimethylphenylsilyl)--->1,3,2-Dioxaborolane, 4,4,5,5-tetramethyl-2-(triphenylsilyl)--->Chlorodimethylphenylsilane-->2-Methoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane-->Dimethylphenylsilane-->2-Isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane | | Preparation Products | Benzene, [dimethyl(2-phenylethyl)silyl]--->Disilane,1,1,1,2,2-pentamethyl-2-phenyl--->3-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine-->3-METHYLTHIOPHENE-2-BORONIC ACID PINACOL ESTER-->1-Methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-indole-->HEPT-6-ENAL-->PHENYLTRIMETHYLSILANE |
|