|
|
| | Avermectin A1a, 5-O-demethyl-22,23-dihydro- Basic information |
| Product Name: | Avermectin A1a, 5-O-demethyl-22,23-dihydro- | | Synonyms: | 22,23-Dihydro-5-O-demethylavermectin A1a;5-O-Demethyl-22,23-dihydroavermectin A1a;Dihydroavermectin B1a;Ivermectin B1a;IvermectinH2B1a;(2Ae,4E,5'S,6S,6'R,7S,8E,11R,13R,15S,17ar,20R,20ar,20bs)-20,20B-dihydroxy-5',6,8,19-tetramethyl-6'-[(1S)-1-methylpropyl]-17-oxo-3',4',5',6,6',10,11,14,15,17,17A,20,20A,20B-tetradecahydro-2H,7H-spiro[11,15-methanofuro[4,3,2-pq][2,6]benzodioxacyclooctadecine-13,2'-pyran]-7-yl 2,6-dideoxy-4-o-(2,6-dideoxy-3-o-methyl-alpha-L-arabino-hexopyranosyl)-3-o-methyl-alpha-L-arabino-hexopyranoside;Avermectin A1a, 5-O-demethyl-22,23-dihydro-;Ivermectin Impurity 3 | | CAS: | 71827-03-7 | | MF: | C48H74O14 | | MW: | 875.09 | | EINECS: | 276-046-2 | | Product Categories: | Organics | | Mol File: | 71827-03-7.mol | 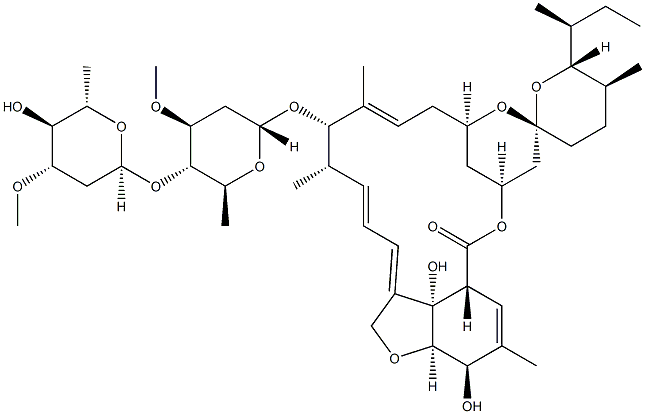 |
| | Avermectin A1a, 5-O-demethyl-22,23-dihydro- Chemical Properties |
| Melting point | 155-157° | | Boiling point | 717.97°C (rough estimate) | | density | 1.0683 (rough estimate) | | refractive index | 1.6130 (estimate) | | storage temp. | Store at -20°C | | solubility | Soluble in DMSO | | form | solid | | pka | 12.42±0.70(Predicted) | | color | white |
| | Avermectin A1a, 5-O-demethyl-22,23-dihydro- Usage And Synthesis |
| Description | Ivermectin B1a is the main component (>80%) of the anthelmintic ivermectin, which also contains ivermectin B1b (<20%; ). It produces antiparasitic activity by binding to glutamate-gated chloride channels expressed on nematode neurons and pharyngeal muscle cells, inducing irreversible channel opening and very long-lasting hyperpolarization/depolarization of the neuron/muscle cell, thereby blocking further function (EC50 = 104 nM). Formulations containing ivermectin inhibit replication of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in Vero/hSLAM cells. | | Uses | Ivermectin B1a-D2 is a deuterium labelled analogue of Ivermectin B1a (I940815), which is a major component of Ivermectin (I940800), a semi-synthetic derivative of Abamectin; consists of a mixture of not less than 80% component B1a and not more than 20% component B1b. Antihelmintic (Onchocerca). An invitro inhibitor of SARS-CoV-2/ Covid-19. | | Definition | ChEBI: A macrocyclic lactone that is avermectin B1a in which the double bond present in the spirocyclic ring system has been reduced to a single bond. It is the major component of ivermectin. | | references | [1] j. wolsetnholme and a. t. rogers. glutamate-gated chloride channels and the mode of action of the avermectin/milbemycin anthelmintics. parasitology131 suppl, s85-s95 (2005).
[2] s. gaisser, l. kellenberger, a. l. kaja, et al. direct production of ivermectin-like drugs after domain exchange in the avermectin polyketide synthase of streptomyces avermitilis atcc31272. organic & biomolecular chemistry 1(16), 2840-2847 (2003).
[3] j. p. arena, k. k. liu, p. s. paress, et al. the mechanism of action of avermectins in caenorhabditis elegans: correlation between activation of glutamate-sensitive chloride current, membrane binding, and biological activity. journal of parasitology 81, 286-294 (1995). |
| | Avermectin A1a, 5-O-demethyl-22,23-dihydro- Preparation Products And Raw materials |
|