|
|
| | 2-isopropyl-5-methylhex-2-en-1-ol Basic information |
| Product Name: | 2-isopropyl-5-methylhex-2-en-1-ol | | Synonyms: | 2-Hexen-1-ol, 5-methyl-2-(1-methylethyl)-;2-ISOPROPYL-5-METHYL-2-HEXENE-1-OL;5-Methyl-2-(1-methylethyl)-2-hexen-1-ol;2-isopropyl-5-methylhex-2-en-1-ol;(E)-5-methyl-2-propan-2-ylhex-2-en-1-ol | | CAS: | 40853-53-0 | | MF: | C10H20O | | MW: | 156.27 | | EINECS: | 255-111-9 | | Product Categories: | | | Mol File: | 40853-53-0.mol | 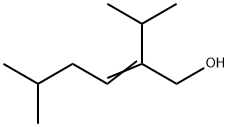 |
| | 2-isopropyl-5-methylhex-2-en-1-ol Chemical Properties |
| Melting point | -4.05°C (estimate) | | Boiling point | 245.54°C (estimate) | | density | 0.8651 (estimate) | | refractive index | 1.4452 (estimate) | | solubility | Very slightly soluble in water, soluble in alcohol and oils. Somewhat soluble in Propylene glycol. | | color | A colourless liquid | | Odor | Herbaceous, minty-Lavender-like taste, but rat her bitter at concentrations higher than 20 ppm. | | Odor Type | herbal | | LogP | 3.291 (est) | | EPA Substance Registry System | 2-Hexen-1-ol, 5-methyl-2-(1-methylethyl)- (40853-53-0) |
| toxicity | Both the acute oral LD50 value in rats and the acute dermal LD50 value in rabbits exceeded 5 g/kg |
| | 2-isopropyl-5-methylhex-2-en-1-ol Usage And Synthesis |
| Occurrence | Has apparently not been reported to occur in nature. | | Uses | This material, developed at the time when Lavandulol was still relatively expensive, offers quite refreshing notes at low cost, but it does not substitute for Lavandulol in artificial Lavender or hvandin oils. 2-isopropyl-5-methylhex-2-en-1-ol finds use in soap and detergent perfumes, room spray fragrances, etc. There is considerable difference in odor between the materials from different suppliers. | | Preparation | By reduction of isodihydrolavandulyl aldehyde |
| | 2-isopropyl-5-methylhex-2-en-1-ol Preparation Products And Raw materials |
|