resDetect™ Kanamycin ELISA Kit
背景(Background)
Residues of kanamycin are prone to occur in the production process of biological products, which can easily lead to abnormal reactions in the human body. Therefore, the residual amount of kanamycin in biological products or semi-finished products of biological products should be strictly controlled.
原理(Assay Principles)
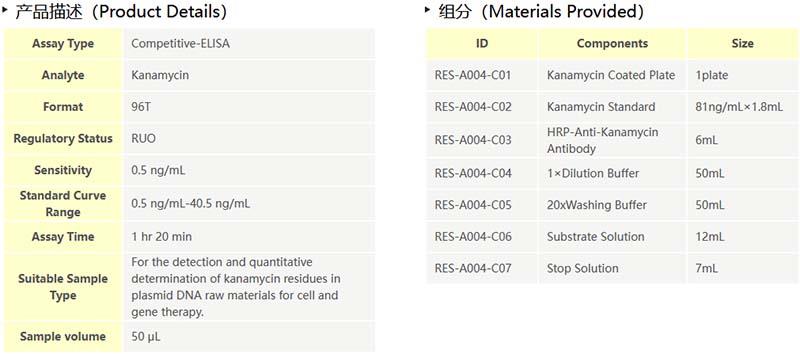
The Kanamycin quantitative detection kit adopts the indirect competitive ELISA method, and the pre-coated conjugated kanamycin antigen on the microstrip competes with the residual kanamycin in the sample to bind the enzyme-labeled anti-kanamycin monoclonal antibody, and then uses a microplate reader to detect the absorbance value by adding TMB substrate, and the absorbance value is negatively correlated with the content of kanamycin in the sample. The kit takes only about one hour and 20 minutes to operate and has a linear range of 0.5 ng/mL to 40.5 ng/mL.
应用说明(Application)
The Kanamycin ELISA Kit is calibrated against the NIFDC standard.
The kit is developed for the detection and quantitative determination of kanamycin residues in plasmid DNA raw materials and proteins for CGT, vaccine and other biological drugs.
It is for research use only.
重构方法(Reconstitution)
Please see Certificate of Analysis for details of reconstitution instruction and specific concentration.
存储(Storage)
1. The kit should be stored at 2°C to 8°C upon receiving.
2. The reconstructed kanamycin standard is stored at -70°C in at least 300 uL per tube and cannot be frozen and thawed repeatedly.
3. Find the expiration date on the outside packaging and do not use reagents past their expiration date.

关键字: Kanamycin ELISA Kit;Kanamycin试剂盒;Kanamycin ELISA试剂盒;ACRO;百普赛斯;
百普赛斯集团ACROBiosystems Group(股票代码:301080)是成立于2010年的跨国生物科技公司,是为全球生物医药、健康产业领域提供关键生物试剂产品及解决方案的行业平台型基石企业。2021年在创业板上市。百普赛斯集团业务遍布全球,横跨亚洲、北美洲、欧洲,在中国、美国、瑞士等12个城市设有办公室、研发中心及生产基地。目前累计服务客户超6000家,与全球Top 20医药企业均建立了长期、稳定的合作伙伴关系。集团旗下拥有品牌ACROBiosystems百普赛斯、bioSeedin柏思荟、Condense Capital垦拓资本和ACRODiagnostics百斯医学等。