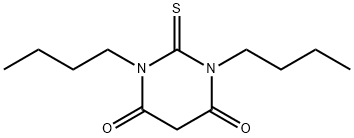
1,3-DI-N-BUTYL-2-THIOBARBITURIC ACID synthesis
- Product Name:1,3-DI-N-BUTYL-2-THIOBARBITURIC ACID
- CAS Number:54443-89-9
- Molecular formula:C12H20N2O2S
- Molecular Weight:256.36
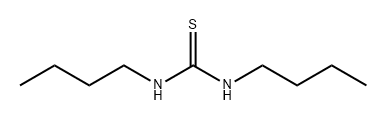
109-46-6
205 suppliers
$16.00/25g
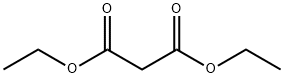
105-53-3
736 suppliers
$5.00/25g

54443-89-9
15 suppliers
$914.40/1G
Yield:54443-89-9 75%
Reaction Conditions:
with sodium ethanolate for 72 h;
Steps:
1 Synthesis of Compound C1: 4,6(1H, 5H)-Pyrimidinedione, 1,3-dibutyldihydro-2-thioxo
The subject compound was synthesised as described generally in Brown (1962) as follows: A 3-necked 2 liter flask fitted with a water condenser, gas inlet, and suba seal was purged with dry N2. A freshly prepared solution of sodium ethyrate, prepared by slowly adding 2.5 g of Na to 50 ml EtOH at room temperature, was added to the flask. Diethyl malonate (17 g, 110 mmol) was then added with vigorous stirring, followed by the addition of N,N'-dibutyl-2-thiourea (10 g, 53 mmol). The mixture was stirred vigorously under N2 for 3 days. The mixture was cooled to room temperature, 50 ml of water was added carefully and ethanol was removed under reduced pressure. The remaining residue was poured into water (200 ml) and cooled in an ice/water bath. The solution was filtered to remove unreacted starting materials and acidified with dilute HCl. The resulting precipitate was collected by suction filtration, washed with water, and dried thoroughly to give a white solid. The yield was about 75%. Compound C1 was determined to be approximately 98% pure by TLC and NMR.
References:
US2007/259903,2007,A1 Location in patent:Page/Page column 5

592-82-5
130 suppliers
$12.00/1g

54443-89-9
15 suppliers
$914.40/1G