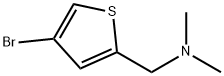
1-(4-BroMothiophen-2-yl)-N,N-diMethylMethanaMine synthesis
- Product Name:1-(4-BroMothiophen-2-yl)-N,N-diMethylMethanaMine
- CAS Number:78909-24-7
- Molecular formula:C7H10BrNS
- Molecular Weight:220.13

18791-75-8
275 suppliers
$9.00/5g
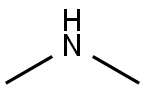
124-40-3
499 suppliers
$18.00/100ml

78909-24-7
16 suppliers
inquiry
Yield:78909-24-7 86%
Reaction Conditions:
with sodium cyanoborohydride;acetic acid in tetrahydrofuran;ethanol at 20;
Steps:
3.a
To a solution of 4-bromothiophene-2-carboxyaldehyde (5g, 26.3 mmol) in EtOH (130 mL) at rt and under N2 was added the dimethylamine in THF (2OmL of a 2.0 M solution, 40 mmol) followed by AcOH (1.9 mL). NaBH3CN (2.1 g, 34 mmol) was then added in portions to the reaction mixture over a 5 min period and the reaction mixture was stirred overnight at rt. The reaction mixture was diluted with saturated NaHCO3 and extracted with CHCl3 (3 x 50 mL). The combined organic layer was concentrated and the resulting residue was diluted with 1 N HCl and washed with CHCl3. The acidic aqueous layer was then made basic with the addition of saturated NaHCO3 and extracted with CHCl3 (3 x 50 mL). The combined organic layer was washed with brine, dried (Na2SO4), filtered and concentrated (86 % yield). The product was used without further purification. 1H NMR (500 MHz, CD3OD) δ 2.26 (s, 6H), 3.65 (s, 2H), 6.92 (apparent s, IH), 7.31 (d, J= 2Hz, IH).
References:
WO2007/133155,2007,A1 Location in patent:Page/Page column 21