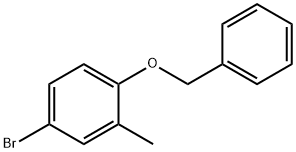
1-(benzyloxy)-4-bromo-2-methylbenzene synthesis
- Product Name:1-(benzyloxy)-4-bromo-2-methylbenzene
- CAS Number:338454-32-3
- Molecular formula:C14H13BrO
- Molecular Weight:277.16

2362-12-1
186 suppliers
$5.00/1g
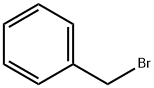
100-39-0
425 suppliers
$10.00/10g

338454-32-3
24 suppliers
$120.00/100mg
Yield:338454-32-3 99%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide at 20; for 72 h;
Steps:
107.1 Step 1 : l-(Benzyloxy)-4-bromo-2-methylbenzene
Example 107 (E)-3 -(4-(6-hvdroxy-2-(4-isobutyl-3 -methylphenyl)- 1 -methyl- 1.2.3.4-tetrahydroisoquinolin- 1 - vDphenvDacrylic acid: Step 1 : l-(Benzyloxy)-4-bromo-2-methylbenzene: 4-Bromo-2-methylphenol (0.5 g, 2.67 mmol) was dissolved in dimethy If orm amide (5.35 ml). Potassium carbonate (0.739 g, 5.35 mmol) was added followed by benzyl bromide (0.350 ml, 2.94 mmol). The reaction was stirred at rt for 72 h. The reaction was quenched with water and extracted with dichloromethane three times. The organic layers were combined, passed through a phase separator and concentrated. The crude material was purfied by silica gel chromatography (0-30% ethyl acetate/heptanes) to afford product (732 mg, 2.64 mmol, 99 % yield) as a clear oil. NMR (400 MHz, Chloroform-d) δ 7.49 - 7.32 (m, 5H), 7.31 (dd, J = 2.6, 0.9 Hz, 1H), 7.26 (ddd, J = 8.6, 2.5, 0.7 Hz, 1H), 6.77 (d, J = 8.6 Hz, 1H), 5.08 (s, 2H), 2.28 (s, 3H).
References:
WO2015/92634,2015,A1 Location in patent:Paragraph 00481

2362-12-1
186 suppliers
$5.00/1g
100-44-7
631 suppliers
$13.50/250G

338454-32-3
24 suppliers
$120.00/100mg