
1-Boc-4-amino-4-piperidineacetic acid synthesis
- Product Name:1-Boc-4-amino-4-piperidineacetic acid
- CAS Number:1159983-30-8
- Molecular formula:C12H22N2O4
- Molecular Weight:258.31

A mixture of 4-oxo-piperidine- 1 -carboxylic acid tert-butyl ester (3.5 g, 17.55 mmol) (Sigma- Aldrich), malonic acid (2.0 g, 19.30 mmol) and ammonium acetate (3.1 g, 40.36 mmol) in n-BuOH was refluxed for 3 h. After cooled, white solid was collected, rinsed with EtOAc and dried in vacuo to provide the title compound 1.61 g (6.2 mmol) in 36% yield.

141-82-2
738 suppliers
$5.00/25g
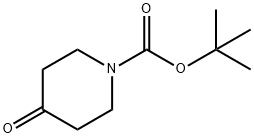
79099-07-3
543 suppliers
$5.00/5g

1159983-30-8
41 suppliers
$52.00/100mg
Yield:1159983-30-8 36%
Reaction Conditions:
with ammonium acetate in butan-1-ol; for 3 h;Reflux;
Steps:
96.a 4-Amino-4-carboxymethyl-piperidine-1-carboxylic acid tert-butyl ester
A mixture of 4-oxo-piperidine- 1 -carboxylic acid tert-butyl ester (3.5 g, 17.55 mmol) (Sigma- Aldrich), malonic acid (2.0 g, 19.30 mmol) and ammonium acetate (3.1 g, 40.36 mmol) in n-BuOH was refluxed for 3 h. After cooled, white solid was collected, rinsed with EtOAc and dried in vacuo to provide the title compound 1.61 g (6.2 mmol) in 36% yield. NMR (200 MHz) in D20, δ ppm: 3.66 (br m, 2 H), 3.10 (br m, 2H), 2.45 (s, 2 H), 1.69 (br m, 4 H), 1.28 (s, 9 H).
References:
WO2013/134660,2013,A1 Location in patent:Paragraph 0373