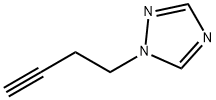
1-(but-3-ynyl)-1H-1,2,4-triazole synthesis
- Product Name:1-(but-3-ynyl)-1H-1,2,4-triazole
- CAS Number:933045-04-6
- Molecular formula:C6H7N3
- Molecular Weight:121.14
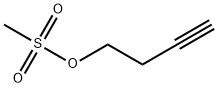
72486-09-0
32 suppliers
inquiry

933045-04-6
4 suppliers
inquiry
Yield:933045-04-6 46%
Reaction Conditions:
Stage #1: 1,2,4-Triazolewith sodium hydroxide;potassium iodide in 2-methyl-butanol, 2-; for 1 h;Heating / reflux;
Stage #2: methanesulfonic acid but-3-ynyl ester in 2-methyl-butanol, 2- at 20; for 3 h;Heating / reflux;
Steps:
2.a
a) l-But-3-ynyl- IH-[1, 2,4] triazoleA mixture of 10.0 g (145 mmol) 1,2,4-triazole, 6.21 g (155 mmol) sodium hydroxide and 17.17 g potassium iodide in 225 ml 2-methyl-butan-2-ol was refluxed for one hour. After cooling down to room temperature 15.33 g (103 mmol) methanesulfonic acid but-3-ynyl ester were added and the mixture was refluxed for three hours. The solvent was removed in vacuo and the residue was dissolved in water and extracted with ethyl acetate several times. The combined organic layers were dried over Na2Sθ4 and the solvent was removed in vacuo. Yield:8.0 g (46%) 1 -but-3-ynyl- IH- [1,2,4] triazole as a yellow liquid.MS: M = 122.1 (ES+)1H-NMR (400 MHz, D^-DMSO): δ= 2.71 (td, 6.7 Hz, 2.6 Hz, 2H), 2.87 (t, 2.6 Hz,IH), 4.31 (t, 6.6 Hz, 2H), 7.98 (s, IH), 8.53 (s, IH).
References:
WO2007/39226,2007,A1 Location in patent:Page/Page column 20