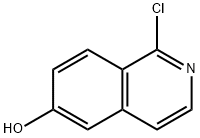
1-chloroisoquinolin-6-ol synthesis
- Product Name:1-chloroisoquinolin-6-ol
- CAS Number:850197-67-0
- Molecular formula:C9H6ClNO
- Molecular Weight:179.6
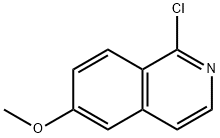
132997-77-4
91 suppliers
$65.00/100mg

850197-67-0
34 suppliers
$233.00/250mg
Yield:-
Steps:
22.22A
22A: 3-(1-chloroisoquinolin-6-yloxy)piperidine-1-carboxylic acid tert-butyl ester The 1-chloro-6-methoxyisoquinoline was prepared as described in WO 00/24718 (Akzo Nobel N.V.). Alternatively it can be prepared from 3-methoxybenzaldehyde using the same procedure as in Example 15A (for the synthesis of 1-chloro-6-methoxy-7-methyl-isoquinoline). The 1-chloro-6-methoxyisoquinoline is demethylated according to the procedure described in WO 00/24718 to afford 1-chloro-6-hydroxyisoquinoline. A suspension of 1-chloro-6-hydroxyisoquinoline (0.18 g, 1 mmol), 3-methanesulfonyloxy-piperidine-1-carboxylic acid tert-butyl ester (336 mg, 1.2 mmol) and 2-tert-butylimino-2-diethylamino-1,3-dimethyl-perhydro-1,3,2-diazaphosphorine on polystyrene (500 mg, 2.2mmol/g loading) in acetonitrile (4 ml) were heated at 120° C. over a period of 900 seconds using the microwave. The excess supported reagent was removed by filtration, washing with methanol, and the filtrate evaporated to dryness under reduced pressure. Since the reaction had not gone to completion, the above procedure was repeated. Purification of the crude material was achieved by chromatography on silica (eluent: 0-25% ethyl acetate in heptane) to afford 3-(1-chloroisoquinolin-6-yloxy)piperidine-1-carboxylic acid tert-butyl ester (106 mg), El-MS: m/z=363.7 [M+H]+.
References:
N.V. Organon US2007/135479, 2007, A1 Location in patent:Page/Page column 12