
1-CYCLOHEXYL-1-TOSYLMETHYL ISOCYANIDE synthesis
- Product Name:1-CYCLOHEXYL-1-TOSYLMETHYL ISOCYANIDE
- CAS Number:1048971-67-0
- Molecular formula:C15H19NO2S
- Molecular Weight:277.38
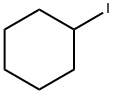
626-62-0
129 suppliers
$10.00/1g
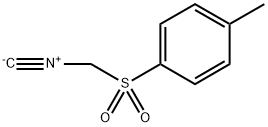
36635-61-7
504 suppliers
$5.00/5g

1048971-67-0
16 suppliers
$1155.00/2.5g
Yield:-
Reaction Conditions:
Stage #1: [(p-methylphenyl)sulfonylmethyl]isonitrilewith sodium hydride in diethyl ether;dimethyl sulfoxide at 0 - 20; for 0.416667 h;
Stage #2: Cyclohexyl iodide in diethyl ether;dimethyl sulfoxide at 20; for 2 h;
Steps:
1.a
NaH (0.8 g, 20.0 mmol, 60% dispersion in oil, 2.0 equiv.) was added to a solution of toluenesulphonylmethyl isocyanide (1.95 g, 10.0 mmol, 1.0 equiv.) in DMSO (10 mL) and Et2O (10 mL) over 5 minutes at 0 C and the reaction mixture was stirred while warming to r.t. for 20 minutes. Cyclohexyl iodide (1.55 mL, 12 mmol, 1.2 equiv) was added dropwise over 3 minutes and the resulting mixture was stirred at r.t. for 2 h. The reaction mixture was then diluted with ethyl acetate (250 mL), and the separated organic layer was washed with water (60 mL), dried over MgSO4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography (20% EtOAc/hexane) to give the product as a white solid: 1H NMR (400MHz, CDCl3) ? 7.88 (d, J= 8.3 Hz, 2 H), 7.43 (d, J = 8.3 Hz, 2 H), 4.30 (d, J= 3.5 Hz, 1 H), 2.50 (s, 3 H), 2.42 (m, 1 H), 2.15 (1 H), 1.85-1.71 (m, 4 H), 1.44-1.19 (m, 5 H).
References:
WO2008/156601,2008,A1 Location in patent:Page/Page column 68