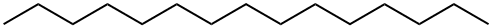
1-PENTADECANOL synthesis
- Product Name:1-PENTADECANOL
- CAS Number:629-76-5
- Molecular formula:C15H32O
- Molecular Weight:228.41

1120-36-1
167 suppliers
$15.00/25mL

107-18-6
2 suppliers
$24.60/100ml
629-76-5
160 suppliers
$48.60/5g
Yield:629-76-5 89 %Chromat.
Reaction Conditions:
with platinum(IV) oxide;Hoveyda-Grubbs catalyst second generation;hydrogen in dichloromethane at 20; under 2250.23 Torr; for 0.833333 h;Inert atmosphere;Autoclave;
Steps:
Condition A: Tandem cross-metathesis/hydrogenation reaction allyl alcohol with 1-octene
General procedure: The primary alcohols with various chain lengths could be obtained by the reaction of allyl alcohol with a terminal olefin using Hoveyda-Grubbs II and PtO2 catalyst together in one pot. The typical procedure for synthesis of 1-nonanol is as follows: a solution of allyl alcohol (0.5 mmol) and 1-octene (1 mmol) in dichloromethane (2.5 mL) was charged into a 10 mL vial followed by Hoveyda-Grubbs II (0.01 mmol) and PtO2 (0.01 mmol) in under an argon atmosphere, and then the via was placed in a 50 mL stainless autoclave. The autoclave was purged with H2 three times and pressurized by H2 (0.3 MPa), and vigorously stirred at room temperature for 50 min.
References:
Jia, Ruilong;Zuo, Zhijun;Li, Xu;Liu, Lei;Dong, Jinxiang [Chinese Chemical Letters,2020,vol. 31,# 6,p. 1525 - 1529] Location in patent:supporting information

6064-48-8
0 suppliers
inquiry
629-76-5
160 suppliers
$48.60/5g

1120-36-1
167 suppliers
$15.00/25mL
201230-82-2
1 suppliers
inquiry
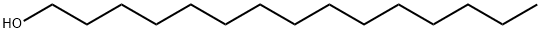
629-76-5
160 suppliers
$48.60/5g
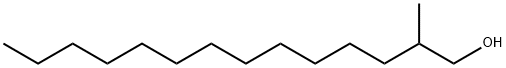
38231-91-3
13 suppliers
inquiry