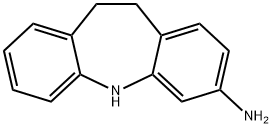
10,11-Dihydro-5H-dibenzo[b,f]azepin-3-aMine synthesis
- Product Name:10,11-Dihydro-5H-dibenzo[b,f]azepin-3-aMine
- CAS Number:10464-35-4
- Molecular formula:C14H14N2
- Molecular Weight:210.27

84803-67-8
64 suppliers
$250.00/1g
![10,11-Dihydro-5H-dibenzo[b,f]azepin-3-aMine](/CAS/20150408/GIF/10464-35-4.gif)
10464-35-4
9 suppliers
inquiry
Yield:10464-35-4 63%
Reaction Conditions:
with hydrogenchloride;acetic acid in water;Reflux;
Steps:
a.iv (iv) SYNTHESIS OF 10,1 1-DIHYDRO-5H-DIBENZO jB,FJAZEPIN-3-AMINE (COMPOUND D)
j00555j Compound C (5.2 g, 0.02 1 mol) in a solution of HC1 (100 mL, 12 mol/L) and AcOH (100 mL) in a 250 mL flask was stirred at reflux for overnight. TLC showed the reaction was completed. The reaction solution was concentrated under reduced pressure and basified with Na2CO3 aqueous solution and extracted with ethyl acetate. The ethyl acetate phase was concentrated under vacuum to give the crude product. The crude product was purified by silica gel chromatography (Petroleum ether/ethyl acetate=5/1) to give compound D(2.2 g, 63%) as abrown solid. LCMS: MS+1=211.1
References:
WO2016/64682,2016,A1 Location in patent:Paragraph 00555

494-19-9
311 suppliers
$6.00/25g
![10,11-Dihydro-5H-dibenzo[b,f]azepin-3-aMine](/CAS/20150408/GIF/10464-35-4.gif)
10464-35-4
9 suppliers
inquiry
13080-75-6
47 suppliers
$150.00/1g
![10,11-Dihydro-5H-dibenzo[b,f]azepin-3-aMine](/CAS/20150408/GIF/10464-35-4.gif)
10464-35-4
9 suppliers
inquiry
![1-(3-Nitro-10,11-dihydro-5H-dibenzo[b,f]azepin-5-yl)ethanone](/CAS/GIF/79752-03-7.gif)
79752-03-7
53 suppliers
inquiry
![10,11-Dihydro-5H-dibenzo[b,f]azepin-3-aMine](/CAS/20150408/GIF/10464-35-4.gif)
10464-35-4
9 suppliers
inquiry