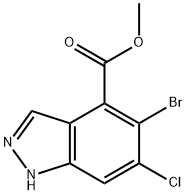
1H-Indazole-4-carboxylic acid, 5-broMo-6-chloro-, Methyl ester synthesis
- Product Name:1H-Indazole-4-carboxylic acid, 5-broMo-6-chloro-, Methyl ester
- CAS Number:1037841-34-1
- Molecular formula:C9H6BrClN2O2
- Molecular Weight:289.51
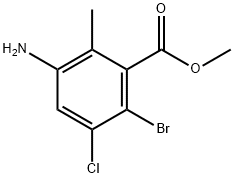
1037841-33-0
5 suppliers
inquiry

1037841-34-1
34 suppliers
$60.00/5mg
Yield:1037841-34-1 76%
Reaction Conditions:
Stage #1: methyl 3-amino-6-bromo-5-chloro-2-methylbenzoatewith acetic acid;sodium nitrite in water at 10 - 30;
Stage #2: with sodium hydrogencarbonate in water;ethyl acetate;
Steps:
56
Reference Example 56; methyl 5-bromo-6-chloro-1H-indazole-4-carboxylate [Show Image]; To a solution of methyl 3-amino-6-bromo-5-chloro-2-methylbenzoate (500 mg, 1.80 mmol) in acetic acid (9 mL) was added a solution of sodium nitrite (137 mg, 1.99 mmol) in water (0.9 mL) at room temperature, and the mixture was stirred for 24 hr. The organic solvent was evaporated under reduced pressure. The residual aqueous solution was diluted with saturated aqueous sodium hydrogen carbonate solution, and the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure, and the residue was purified by silica gel column chromatography (NH, ethyl acetate) to give the title compound (398 mg, yield 76%). 1H-NMR (CDCl3) δ: 4.05 (3H, s), 7.66 (1H, s), 8.31 (1H, s), 10.60 (1H, brs), MS (ESI+): 289 (M+H).
References:
EP2098513,2009,A1 Location in patent:Page/Page column 67