
(3-endo)-benzyl3-(hydroxymethyl)-8-azabicyclo[3.2.1]octane-8-carboxylate synthesis
- Product Name:(3-endo)-benzyl3-(hydroxymethyl)-8-azabicyclo[3.2.1]octane-8-carboxylate
- CAS Number:1067230-68-5
- Molecular formula:C16H21NO3
- Molecular Weight:275.34
![(1R,3r,5S)-8-Azabicyclo[3.2.1]octan-3-ylmethanol](/CAS/20200119/GIF/1257512-65-4.gif)
1257512-65-4
3 suppliers
inquiry
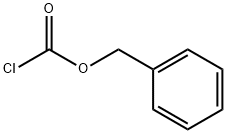
501-53-1
393 suppliers
$10.00/1g
![(3-endo)-benzyl3-(hydroxymethyl)-8-azabicyclo[3.2.1]octane-8-carboxylate](/CAS/20180702/GIF/1067230-68-5.gif)
1067230-68-5
14 suppliers
$150.00/50mg
Yield:1067230-68-5 61%
Reaction Conditions:
with triethylamine in acetonitrile at 0 - 20; for 2 h;
Steps:
29.4
TEA (4.4 mL, 31.65 mmol) was added to a suspension of 100a (2.98 g, 21.103 mmol) in MeCN (70 mL) and the reaction flask was cooled to 0° C. Benzyl chloroformate (4.8 mL, 31.65 mmol) was added to the solution and the reaction mix was allowed to warm up to RT and stirred for 2 h. The residue was partitioned between water and DCM and the aqueous layer was back extracted twice with DCM. The combined extract were dried (Na2SO4), filtered and evaporated. The remaining orange oil was purified by SiO2 chromatography eluting with a MeOH/DCM gradient (0 to 4% MeOH) to afford 3.57 g (61%) of 100b as a light yellow oil.
References:
US2008/249087,2008,A1 Location in patent:Page/Page column 33