
2-(4-ethylcyclohex-1-enyl)-4,4,5,5-tetraMethyl-1,3,2-dioxaborolane synthesis
- Product Name:2-(4-ethylcyclohex-1-enyl)-4,4,5,5-tetraMethyl-1,3,2-dioxaborolane
- CAS Number:1092938-90-3
- Molecular formula:C14H25BO2
- Molecular Weight:236.16

173322-26-4
0 suppliers
inquiry
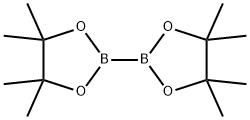
73183-34-3
573 suppliers
$6.00/5g

1092938-90-3
52 suppliers
$45.00/50mg
Yield:1092938-90-3 60%
Reaction Conditions:
with potassium carbonate;bis-triphenylphosphine-palladium(II) chloride;triphenylphosphine in 1,4-dioxane at 80;Inert atmosphere;
Steps:
24A
Example 24A 2-(4-Ethylcyclohex-1-en-1-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane Under an atmosphere of argon, 1180 mg (4.57 mmol) of 4-ethylcyclohex-1-en-1-yl trifluoromethanesulfonate, 1276 g (5.03 mmol) of 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi-1,3,2-dioxaborolane, 947 mg (6.85 mmol) of potassium carbonate, 72 mg (0.27 mmol) of triphenylphosphine and 96 mg (0.14 mmol) of bis(triphenylphosphine)palladium(II) chloride are initially charged in 3 ml of dioxane. The reaction mixture is stirred at 80° C. overnight. The reaction mixture is diluted with dichloromethane and washed with water and saturated sodium chloride solution, and the aqueous phase is extracted with dichloromethane. The combined organic phases are dried over sodium sulfate and concentrated on a rotary evaporator. The residue is dried under reduced pressure and chromatographed on silica gel using a mobile phase of cyclohexane and ethyl acetate 10/1. This gives 691 mg (60% of theory) of the target compound. GC-MS (method 9): Rt=4.78 min; m/z=236 (M)+. 1H-NMR (400 MHz, DMSO-d6): δ=6.42 (m, 1H), 2.21-2.05 (m, 2H), 2.01-1.89 (m, 1H), 1.72-1.58 (m, 2H), 1.42-1.12 (m, 16H: including 1.25 (t, 2H) and 1.18 (s, 12H)).
References:
US2011/166163,2011,A1 Location in patent:Page/Page column 27