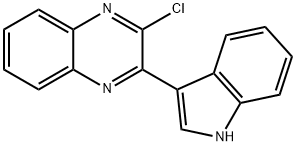
2-Chloro-3-(1H-indol-3-yl)-quinoxaline, 98+% C16H10ClN3, MW: 279.73 synthesis
- Product Name:2-Chloro-3-(1H-indol-3-yl)-quinoxaline, 98+% C16H10ClN3, MW: 279.73
- CAS Number:1146080-41-2
- Molecular formula:C16H10ClN3
- Molecular Weight:279.72
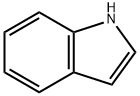
120-72-9
620 suppliers
$6.00/25g
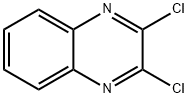
2213-63-0
340 suppliers
$11.00/5g

1146080-41-2
13 suppliers
$599.00/1g
Yield:1146080-41-2 85%
Reaction Conditions:
with aluminum (III) chloride in 1,2-dichloro-ethane at 80; for 0.5 h;Inert atmosphere;
Steps:
4.1.2. General procedure for the preparation of compound 3
General procedure: A mixture of 2,3-dichloroquinoxaline (1, 1.0 equiv), an appropriate (hetero)arene (2, 1.0 equiv) and AlCl3 (1.1 equiv) in dichloroethane (5 mL) was stirred at 80 °C for time indicated in Table 2 under a nitrogen atmosphere. After completion of the reaction, the mixture was poured into ice-cold water (15 mL), stirred for 10 min and then extracted with ethylacetate (3 × 20 mL). The organic layers were collected, combined, washed with cold water (2 × 20 mL), dried over anhydrous Na2SO4 and concentrated under vacuum. The residue obtained was purified by column chromatography using ethylacetate/hexene to give the desired product.
References:
Kumar, K. Shiva;Rambabu;Sandra, Sandhya;Kapavarapu, Ravikumar;Krishna, G. Rama;Basaveswara Rao;Chatti, Kiranam;Reddy, C. Malla;Misra, Parimal;Pal, Manojit [Bioorganic and Medicinal Chemistry,2012,vol. 20,# 5,p. 1711 - 1722] Location in patent:experimental part