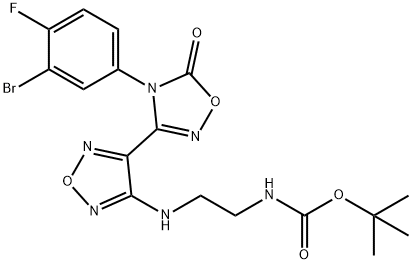
Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester synthesis
- Product Name:Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester
- CAS Number:1204669-68-0
- Molecular formula:C17H18BrFN6O5
- Molecular Weight:485.26

24424-99-5
858 suppliers
$13.50/25G
![1,2,4-Oxadiazol-5(4H)-one, 3-[4-[(2-azidoethyl)amino]-1,2,5-oxadiazol-3-yl]-4-(3-bromo-4-fluorophenyl)-](/CAS/20180703/GIF/1204669-66-8.gif)
1204669-66-8
0 suppliers
inquiry
![Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester](/CAS/20200119/GIF/1204669-68-0.gif)
1204669-68-0
6 suppliers
inquiry
Yield:1204669-68-0 83%
Reaction Conditions:
Stage #1: 3-(4-((2-azidoethyl)amino)-1,2,5-oxadiazole-3-yl)-4-(3-bromo-4-fluorophenyl)-1,2,4-oxadiazole-5(4H)-onewith methanol;sodium iodide for 0.5 h;Large scale;
Stage #2: with chloro-trimethyl-silane in methanol at 20 - 35; for 2.5 h;Large scale;
Stage #3: di-tert-butyl dicarbonatewith potassium carbonate in methanol;water at 20; pH=9;Large scale;
Steps:
1.11 Step 11: 3-{4-[(2-Aminoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)- 1.2.4- oxadiazol-5(4H)-one hydrochloride
Step 11: 3-{4-[(2-Aminoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)- 1.2.4- oxadiazol-5(4H)-one hydrochloride [00194] Sodium iodide (1080 g, 7.2 mol) was added to 3-{4-[(2-azidoethyl)amino]- 1.2.5- oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)-l ,2,4-oxadiazol-5(4H)-one (500 g, 1.22 mol) in methanol (6 L). The mixture was allowed to stir for 30 min during which time a mild exotherm was observed. Chlorotrimethylsilane (930 mL, 7.33 mol) was added as a solution in methanol (1 L) dropwise at a rate so that the temperature did not exceed 35 °C, and the reaction was allowed to stir for 3.5 h at ambient temperature. The reaction was neutralized with 33 wt% solution of sodium thiosulfate pentahydrate in water (-1.5 L), diluted with water (4 L), and the pH adjusted to 9 carefully with solid potassium carbonate (250 g - added in small portions: watch foaming). Di-ieri-butyl dicarbonate (318 g, 1.45 mol) was added and the reaction was allowed to stir at room temperature. Additional potassium carbonate (200 g) was added in 50 g portions over 4 h to ensure that the pH was still at or above 9. After stirring at room temperature overnight, the solid was filtered, triturated with water (2 L), and then MTBE (1.5 L). A total of 11 runs were performed (5.5 kg, 13.38 mol). The combined solids were triturated with 1 : 1 THF:dichloromethane (24 L, 4 runs in a 20 L rotary evaporator flask, 50 °C, 1 h), filtered, and washed with dichloromethane (3 L each run) to afford an off- white solid. The crude material was dissolved at 55 °C tetrahydrofuran (5 mL/g), treated with decolorizing carbon (2 wt%) and silica gel (2 wt%), and filtered hot through celite to afford the product as an off-white solid (5122 g). The combined MTBE, THF, and dichloromethane filtrates were concentrated in vacuo and chromatographed (2 kg silica gel, heptane with a 0-100% ethyl acetate gradient, 30 L) to afford more product (262 g). The combined solids were dried to a constant weight in a convection oven (5385 g, 83%). [00195] In a 22 L flask was charged hydrogen chloride (4 N solution in 1 ,4-dioxane, 4 L, 16 mol). tert-Butyl [2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l ,2,4- oxadiazol-3-yl]-l ,2,5-oxadiazol-3-yl}amino)ethyl]carbamate (2315 g, 4.77 mol) was added as a solid in portions over 10 min. The slurry was stirred at room temperature and gradually became a thick paste that could not be stirred. After sitting overnight at room temperature, the paste was slurried in ethyl acetate (10 L), filtered, re-slurried in ethyl acetate (5 L), filtered, and dried to a constant weight to afford the desired product as a white solid (combined with other runs, 5 kg starting material charged, 41 13 g, 95%). LCMS for Ci2HnBrFN603 (M+H)+: m/z = 384.9, 386.9. 1H NMR (400 MHz, DMSO-^): δ 8.12 (m, 4 H), 7.76 (m, 1 H), 7.58 (t, J = 8.7 Hz, 1 H), 6.78 (t, J = 6.1 Hz, 1 H), 3.51 (dd, J = 1 1.8, 6.1 Hz, 2 H), 3.02 (m, 2 H).
References:
WO2014/66834,2014,A1 Location in patent:Paragraph 00194-00195; 00304

914471-43-5
41 suppliers
inquiry

89711-08-0
171 suppliers
$27.00/250mg
![Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester](/CAS/20200119/GIF/1204669-68-0.gif)
1204669-68-0
6 suppliers
inquiry
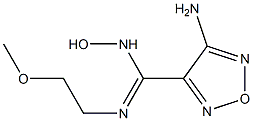
1204669-59-9
0 suppliers
inquiry
![Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester](/CAS/20200119/GIF/1204669-68-0.gif)
1204669-68-0
6 suppliers
inquiry
![1,2,5-Oxadiazole-3-carboximidamide,N-hydroxy-4-[(2-methoxyethyl)amino]-](/CAS/20180702/GIF/1204669-60-2.gif)
1204669-60-2
0 suppliers
inquiry
![Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester](/CAS/20200119/GIF/1204669-68-0.gif)
1204669-68-0
6 suppliers
inquiry
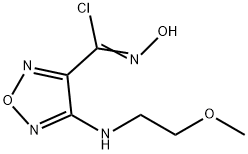
1204669-61-3
12 suppliers
$45.00/2.5mg
![Carbamic acid,N-[2-[[4-[4-(3-bromo-4-fluorophenyl)-4,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl]-1,2,5-oxadiazol-3-yl]amino]ethyl]-, 1,1-dimethylethyl ester](/CAS/20200119/GIF/1204669-68-0.gif)
1204669-68-0
6 suppliers
inquiry