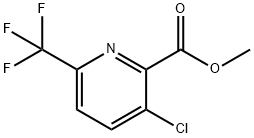
3-Chloro-6-trifluoromethyl-pyridine-2-carboxylic acid methyl ester synthesis
- Product Name:3-Chloro-6-trifluoromethyl-pyridine-2-carboxylic acid methyl ester
- CAS Number:1214324-33-0
- Molecular formula:C8H5ClF3NO2
- Molecular Weight:239.58
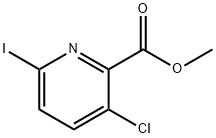
1245735-42-5
2 suppliers
inquiry
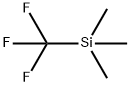
81290-20-2
403 suppliers
$13.00/1g

1214324-33-0
16 suppliers
inquiry
Yield:-
Reaction Conditions:
with potassium fluoride;copper(l) iodide in 1-methyl-pyrrolidin-2-one at 50; for 16 h;Inert atmosphere;
Steps:
Step 3: Into a 3000-mL 4-necked round-bottom flask, purged and maintained withan inert atmosphere of nitrogen, was placed NMP (1800 mL), potassium fluoride (46.9 g, 807.27mmol), copper (I) iodide (164 g, 861.12 mmol) and trimethyl(trifluoromethyl)silane (1 02 mL,1.20 equiv). To this was added methyl3-chloro-6-iodopyridine-2-carboxylate (160 g, 537.8520 mmol) at 50 °C. The resulting solution was stirred for 16 h at 50 °C. The reaction mixture wasquenched by the addition of water/ice. The resulting solution was extracted with ethyl acetate (2x 1000 mL) and the organic layers were combined, dried over anhydrous sodium sulfate andconcentrated in vacuo. This resulted in methyl 3-chloro-6-(trifluoromethyl)pyridine-2-carboxylate as a yellow solid.
References:
WO2014/99836,2014,A1 Location in patent:Page/Page column 32; 33
1702-17-6
314 suppliers
$10.00/5g

1214324-33-0
16 suppliers
inquiry
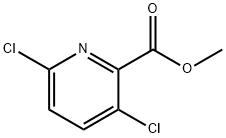
1532-24-7
71 suppliers
$22.00/250mg

1214324-33-0
16 suppliers
inquiry