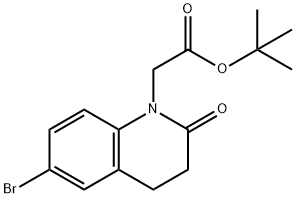
tert-butyl 2-(6-bromo-2-oxo-3,4-dihydroquinolin-1(2H)-yl)acetate synthesis
- Product Name:tert-butyl 2-(6-bromo-2-oxo-3,4-dihydroquinolin-1(2H)-yl)acetate
- CAS Number:1224927-63-2
- Molecular formula:C15H18BrNO3
- Molecular Weight:340.21
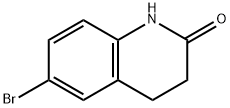
3279-90-1
152 suppliers
$10.00/1g
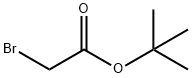
5292-43-3
445 suppliers
$10.00/5g

1224927-63-2
27 suppliers
$13.00/100mg
Yield:1224927-63-2 72%
Reaction Conditions:
Stage #1: 6-bromo-3,4-dihydro-1H-quinolin-2-onewith sodium hydride in N,N-dimethyl-formamide; for 0.5 h;Inert atmosphere;
Stage #2: bromoacetic acid tert-butyl ester in N,N-dimethyl-formamide at 20;
Steps:
3.A
A. Synthesis of tert-butyl 2-(6-bromo-2-oxo-3,4-dihydroquinolin-1(2H)-yl)acetate To a mixture of 95% dry sodium hydride (834 mg, 33.0 mmol) in anhydrous N,N-dimethylformamide (30 mL) at room temperature was added a solution of 6-bromo-3,4-dihydroquinolin-2(1H)-one (6.780 g, 30.00 mmol) in anhydrous N,N-dimethylformamide (10 mL). The reaction mixture was stirred for 30 minutes under an atmosphere of dry N2, followed by addition of a solution of tert-butyl 2-bromoacetate (7.5 mL, 49.7 mmol) in N,N-dimethylformamide (10 mL). The reaction mixture was stirred at room temperature until the majority of the starting material was converted (confirmed by LCMS). The reaction mixture was quenched with methanol (40 mL), the mixture concentrated under reduced pressure, then diluted with ethyl acetate (150 mL). The organic phase was washed with water (100 mL), 30% ammonium chloride (100 mL) and brine (100 mL), dried, and concentrated under reduced pressure. Ethyl ether (20 mL) was added, and the mixture sonicated, filtered, washed with ether (20 mL), and dried to afford tert-butyl 2-(6-bromo-2-oxo-3,4-dihydroquinolin-1(2H)-yl)acetate (7.348 g, 21.6 mmol, 72%). LCMS mz 285.9 (M-56+H), 363.9 (M+Na), anal HPLC>97% in purity. 1H NMR (400 MHz; CDCl3) δ 7.30-7.40 (m, 2H); 7.50-6.70 (m, 1H); 4.54 (s, 2H); 2.92 (m, 2H); 2.69 (m, 2H), 1.44 (s, 9H).
References:
US2010/113514,2010,A1 Location in patent:Page/Page column 17