
benzyl 3,4-dihydro-4-oxo-2-phenylpyridine-1(2H)-carboxylate synthesis
- Product Name:benzyl 3,4-dihydro-4-oxo-2-phenylpyridine-1(2H)-carboxylate
- CAS Number:126378-73-2
- Molecular formula:C19H17NO3
- Molecular Weight:307.34
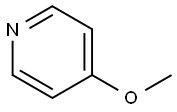
620-08-6
304 suppliers
$5.00/1g
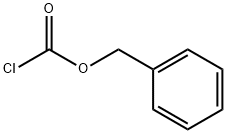
501-53-1
394 suppliers
$10.00/1g
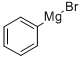
100-58-3
185 suppliers
$18.00/10ml

126378-73-2
13 suppliers
$370.00/1g
Yield:126378-73-2 99.4%
Reaction Conditions:
Stage #1: 4-methoxypyridine;benzyl chloroformate in tetrahydrofuran at -78; for 0.5 h;
Stage #2: phenylmagnesium bromide in tetrahydrofuran at -78 - 20; for 1 h;
Steps:
28.1 Benzyl 4-oxo-2-phenyl-3,4-dihydropyridine-1(2H)-carboxylate
To a solution of 4-methoxypyridine (10.0 g, 91.6 mmol) in THF (500 mL) was added dropwise benzyl chloroformate (15.6 g, 91.6 mmol) at -78 °C and stirred for 30 mm. Then phenylmagnesiumbromide in THF (110 mL, 2M, 110 mmol) was added slowly to the mixture at -78°C. The mixture was stirred at -78 °C for 30 mm, warmed up to room temperature and stirred for 30 mm. Then it was quenched by HCl (2M, 400 mL) and the organic solvent was removed under reduced pressure. The residue was filtered to afford the desired product (28 g, 99.4%) as a white solid. LCMS (ESI) mz: 330.1 [M+Na]+.
References:
WO2017/58821,2017,A1 Location in patent:Page/Page column 123; 124

620-08-6
304 suppliers
$5.00/1g

126378-73-2
13 suppliers
$370.00/1g

501-53-1
394 suppliers
$10.00/1g

126378-73-2
13 suppliers
$370.00/1g