
4H-benzo[e][1,2,4]triazolo[5,1-c][1,2,4]thiadiazine-2-carboxylic acid 5,5-dioxide synthesis
- Product Name:4H-benzo[e][1,2,4]triazolo[5,1-c][1,2,4]thiadiazine-2-carboxylic acid 5,5-dioxide
- CAS Number:1311316-61-6
- Molecular formula:C9H6N4O4S
- Molecular Weight:266.23
![methyl 8,8-dioxo-8lambda6-thia-2,3,5,7-tetraazatricyclo[7.4.0.0,2,6]trideca-1(13),3,5,9,11-pentaene-4-carboxylate](/CAS/20210111/GIF/1311315-62-4.gif)
1311315-62-4
6 suppliers
inquiry
![4H-benzo[e][1,2,4]triazolo[5,1-c][1,2,4]thiadiazine-2-carboxylic acid 5,5-dioxide](/CAS/20200331/GIF/1311316-61-6.gif)
1311316-61-6
11 suppliers
inquiry
Yield:1311316-61-6 98%
Reaction Conditions:
with water;lithium hydroxide at 20;
Steps:
4Н-[1,24]triazolo[5,1-с][1,2,4]benzothiadiazine-2-carboxylate 5,5-dioxide 8
A mixture of methyl-4Н-[1,24]triazolo[5,1-с][1,2,4]benzothiadiazine-2-carboxylate 5,5-dioxide 6a (1.4 g, 0.005 mol), lithium hydroxide (0.264 g, 0.011 mol), and water (50 mL) was stirred at room temperature overnight. The reaction mixture was acidified to pH 1 with concentrated hydrochloric acid. The precipitate was filtered, washed with water (3x50 mL), and dried in vacuum. Yield 1.3 g (98 %).
References:
Cherepakha, Artem;Kovtunenko, Vladimir O.;Tolmachev, Andrey [Tetrahedron Letters,2013,vol. 54,# 8,p. 986 - 988] Location in patent:supporting information
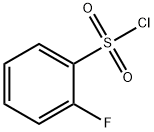
2905-21-7
220 suppliers
$9.00/5g
![4H-benzo[e][1,2,4]triazolo[5,1-c][1,2,4]thiadiazine-2-carboxylic acid 5,5-dioxide](/CAS/20200331/GIF/1311316-61-6.gif)
1311316-61-6
11 suppliers
inquiry