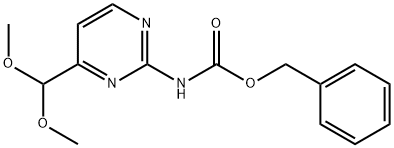
Benzyl (4-(Dimethoxymethyl)Pyrimidin-2-Yl)Carbamate synthesis
- Product Name:Benzyl (4-(Dimethoxymethyl)Pyrimidin-2-Yl)Carbamate
- CAS Number:1312764-25-2
- Molecular formula:C15H17N3O4
- Molecular Weight:303.31
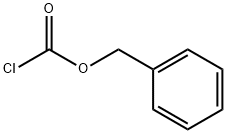
501-53-1
396 suppliers
$10.00/1g
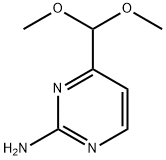
165807-05-6
105 suppliers
$19.00/100mg

1312764-25-2
14 suppliers
$421.00/1g
Yield:1312764-25-2 90%
Reaction Conditions:
Stage #1: 4-(dimethoxymethyl)pyrimidin-2-aminewith sodium hydrogencarbonate in ethanol;water; for 0.25 h;
Stage #2: benzyl chloroformate in 1,4-dioxane;ethanol;water at 5 - 10; for 12 h;
Steps:
Benzyl-(4-(dimethoxymethyl)pyrimidin-2-yl)carbamate (3)
4-(Dimethoxymethyl) pyrimidin-2-amine (2) (10 g, 59 mmol) was dissolved in small amount of ethanol (15 ml) and stirred for 10-15 min. Solution of NaHCO3 (9.9 g, 118 mmol) in water was then added to it at room temperature. After 15 min of stirring, solution of CbzCl (16.8 ml, 118 mmol) in dioxane (20 ml) was added drop wise at cold condition. The reaction mixture was then warmed to room temperature gradually and stirred overnight. Ethanol was concentrated on rotavapor and water was added. The aqueous layer was extracted with ethyl acetate to remove any organic impurities. The organic layer was back extracted with saturated NaHCO3 solution. The combined aqueous layers were acidified with conc. HCl and then extracted with ethyl acetate. The organic layer was then concentrated and purified over column chromatography (ethyl acetate-hexane, 30:70) to yield white crystalline solid 3 (16 g, 90%).
References:
Naik, Nilesh H.;Sikder, Arun K.;Kusurkar, Radhika S. [Tetrahedron Letters,2013,vol. 54,# 28,p. 3715 - 3717] Location in patent:supporting information
6342-56-9
307 suppliers
$8.00/25g

1312764-25-2
14 suppliers
$421.00/1g