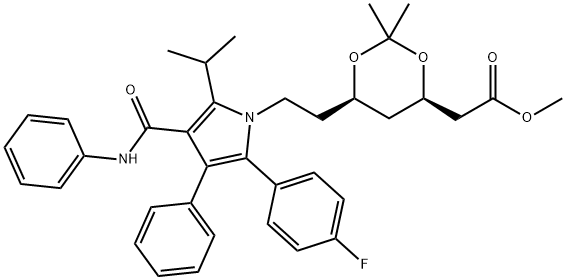
methyl 2-((4R,6R)-6-(2-(2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4- (phenylcarbamoyl)-1H-pyrrol-1-yl)ethyl)-2,2-dimethyl-1,3-dioxan-4- yl)acetate synthesis
- Product Name:methyl 2-((4R,6R)-6-(2-(2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4- (phenylcarbamoyl)-1H-pyrrol-1-yl)ethyl)-2,2-dimethyl-1,3-dioxan-4- yl)acetate
- CAS Number:1353049-81-6
- Molecular formula:C37H41FN2O5
- Molecular Weight:612.73

77-76-9
390 suppliers
$9.00/5ml
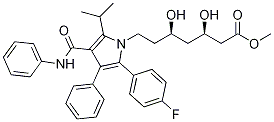
345891-62-5
103 suppliers
$85.00/1mg

1353049-81-6
22 suppliers
inquiry
Yield:1353049-81-6 96%
Reaction Conditions:
with toluene-4-sulfonic acid in dichloromethane; for 1.5 h;Reflux;
Steps:
Methyl 2-((4R,6R)-6-(2-(2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)-1H-pyrrol-1-yl)ethyl)-2,2-dimethyl-1,3-dioxan-4-yl)acetate (3)
2,2-Dimethoxypropane (1.3 mL, 1108 mg, 10.639 mmol) was added to a solution of 2a (509 mg, 0.889 mmol), p-toluenesulfonic acid monohydrate (17 mg, 0.089 mmol) in dichloromethane (13 mL). This solution was stirred for 1.5 h at reflux, and then 10% NaHCO3 aq. (5 mL) was added to quench the reaction. The resulting mixture was extracted with dichloromethane. The organic layer was washed with saturated brine, dried (MgSO4), and evaporated. The crude product was purified using silica gel chromatography (ethyl acetate/n-hexane=1/2) to provide 3 (522 mg, 96% yield) as colorless solids; mp 63-66 °C; IR nmax (KBr) cm-1: 3411, 1739, 1668; 1H-NMR (400 MHz, CDCl3) d: 7.21-7.15 (m, 9H), 7.06 (d, 2H, J=8.0 Hz), 7.01-6.96 (m, 3H), 6.85 (br s, 1H), 4.24-4.17 (m, 1H), 4.11-4.03 (m, 1H), 3.85-3.78 (m, 1H), 3.72-3.67 (m, 4H), 3.62-3.49 (m, 1H), 2.51 (dd, 1H, J=16.0, 6.8 Hz), 2.32 (dd, 1H, J=16.0, 6.8 Hz), 1.72-1.61 (m, 2H), 1.52 (d, 6H, J=7.2 Hz), 1.38-1.35 (m, 4H), 1.30 (s, 3H), 1.04 (dd, 1H, J=24.0, 11.6 Hz); 13C-NMR (100 MHz, CDCl3) d: 171.2, 164.8, 162.2 (d, 1JC-F=246.2 Hz), 141.4, 138.4, 134.6, 133.2 (d, 3JC-F=8.1 Hz), 130.5, 128.7, 128.6, 128.3, 128.2 (d, 4JC-F=3.5 Hz), 126.5, 123.5, 121.7, 119.5, 115.37 (d, 2JC-F=21.3 Hz), 115.35, 98.8, 66.3, 65.5, 51.6, 41.0, 40.8, 38.0, 35.9, 29.9, 26.1, 21.7, 21.5, 19.6; HRMS (ESI) m/z: calcd C37H41FN2NaO5 (M+Na+) 635.2897, found 635.2897.
References:
Mizoi, Kenta;Takahashi, Masato;Haba, Masami;Hosokawa, Masakiyo [Bioorganic and Medicinal Chemistry Letters,2016,vol. 26,# 3,p. 921 - 923] Location in patent:supporting information

67-56-1
740 suppliers
$9.00/25ml
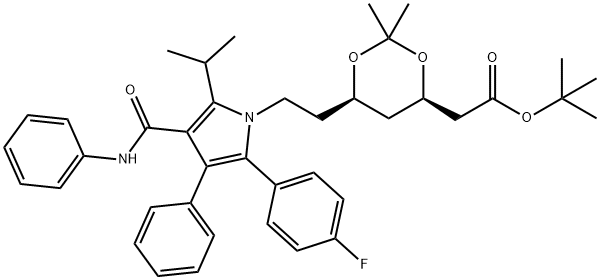
125971-95-1
334 suppliers
$32.00/1g

1353049-81-6
22 suppliers
inquiry
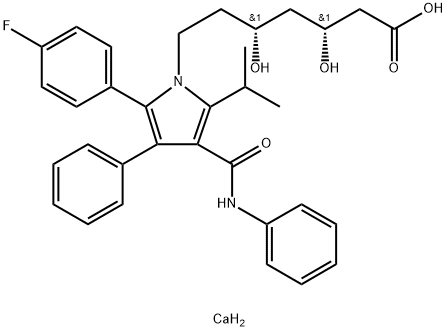
134523-03-8
602 suppliers
$5.00/10mg

1353049-81-6
22 suppliers
inquiry