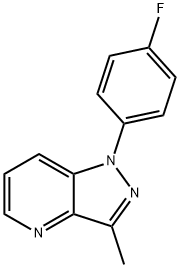
1H-Pyrazolo[4,3-b]pyridine, 1-(4-fluorophenyl)-3-methyl- synthesis
- Product Name:1H-Pyrazolo[4,3-b]pyridine, 1-(4-fluorophenyl)-3-methyl-
- CAS Number:1383734-85-7
- Molecular formula:C13H10FN3
- Molecular Weight:227.24
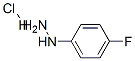
823-85-8
367 suppliers
$5.00/5g

87674-20-2
126 suppliers
$11.00/250mg
![1H-Pyrazolo[4,3-b]pyridine, 1-(4-fluorophenyl)-3-methyl-](/CAS/20210111/GIF/1383734-85-7.gif)
1383734-85-7
0 suppliers
inquiry
Yield:1383734-85-7 84%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide at 120; for 24 h;
Steps:
11.a
a) To a 100-mL roundbottomed flask was added 4-fluorophenylhydrazine hydrochloride (3.33 g, 20.5 mmol), K2CO3 (8.29 g, 60 mmol), and DMF (27 mL). To this slurry was added 2.78 g (20 mmol) of 1-(3-fluoropyridin-2-yl)ethanone, giving a yellow suspension. The mixture was heated to 120° C. for 24 hours at which point the starting materials were consumed. The slurry was cooled to room temperature and 70 mL of H2O was added to precipitate the product. The solid was collected on a medium fit and washed with 3×H2O. The solid was dried at 60° C. to provide the product as a tan solid (3.82 g, 84%). MS: (ES) m/z calculated for C13H11FN3 [M+H]+ 228.1, found 228.1.
References:
US2014/57937,2014,A1 Location in patent:Paragraph 0123; 0124
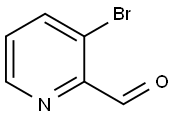
405174-97-2
147 suppliers
$19.00/100mg
![1H-Pyrazolo[4,3-b]pyridine, 1-(4-fluorophenyl)-3-methyl-](/CAS/20210111/GIF/1383734-85-7.gif)
1383734-85-7
0 suppliers
inquiry
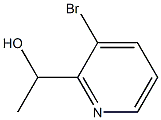
1383734-83-5
13 suppliers
inquiry
![1H-Pyrazolo[4,3-b]pyridine, 1-(4-fluorophenyl)-3-methyl-](/CAS/20210111/GIF/1383734-85-7.gif)
1383734-85-7
0 suppliers
inquiry