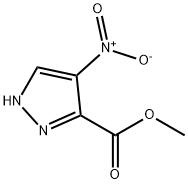
1H-Pyrazole-3-carboxylic acid, 4-nitro-, methyl ester synthesis
- Product Name:1H-Pyrazole-3-carboxylic acid, 4-nitro-, methyl ester
- CAS Number:138786-86-4
- Molecular formula:C5H5N3O4
- Molecular Weight:171.11

67-56-1
740 suppliers
$7.29/5ml-f
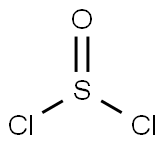
7719-09-7
391 suppliers
$15.00/10g
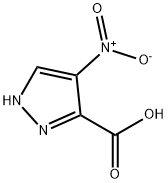
5334-40-7
210 suppliers
$14.14/5gm:

138786-86-4
103 suppliers
$7.00/250mg
Yield:138786-86-4 99.5%
Reaction Conditions:
at 0 - 25; for 16 - 48 h;Product distribution / selectivity;
Steps:
4.1; 11.1
A 2OL reaction vessel equipped with a digital thermometer and stirrer was charged with A- nitro-1 H-pyrazole-3-carboxylic acid (1.117 Kg, 7.1 1 mol, 1 wt) and methanol (8.950 L, 8 vol). The reaction mixture was stirred under nitrogen, cooled to O to 5 0C, thionyl chloride (0.581 L, 8.0 mol, 0.52 vol) added over 180 minutes and the resultant mixture allowed to warm to and stir at 18 to 22 0C overnight, after which time 1H NMR analysis (d6-DMSO) indicated reaction completion. The reaction mixture was concentrated under reduced pressure at 40 to 45 0C, the residue treated with toluene and re-concentrated (3x 2.250 L, 3x 2vol) under reduced pressure at 40 to 45 0C to give 4-nitro-1H-pyrazole-3-carboxylic acid methyl ester as an off-white solid (1.210 Kg, 99.5%).; Thionyl chloride (2.90 ml, 39.8 mmol) was slowly added to a mixture of 4-nitro-3- pyrazolecarboxylic acid (5.68 g, 36.2 mmol) in MeOH (100 ml) at ambient temperature and the mixture stirred for 48 hours. The mixture was reduced in vacuo and dried through azeotrope with toluene to afford 4-nitro-1H-pyrazole-3-carboxylic acid methyl ester as a white solid.; Stage 1 : Preparation of 4-nitro-1 H-pvrazole-3-carboxylic acid methyl ester 4-Nitro-1H-pyrazole-3-carboxylic acid (1.350Kg1 8.59 MoI, 1.0 wt) and methanol (10.80L, 8.0 vol) were charged to a flange flask equipped with a mechanical stirrer, condenser and thermometer. The suspension was cooled to O to 5°C under nitrogen and thionyl chloride (0.702L, 9.62 MoI, 0.52 vol) added at this temperature. The mixture was warmed to 15 to 25°C over 16 to 24 hours. Reaction completion was determined by 1H NMR analysis (d6- DMSO). The mixture was concentrated under vacuum at 35 to 45°C and toluene (2.70L, 2.0 vol) charged to the residue and removed under vacuum at 35 to 45°C. The toluene azeotrope was repeated twice using toluene (2.70L, 2.0 vol) to give 4-nitro-1 H-pyrazole-3- carboxylic acid methyl ester [1.467Kg, 99.8%th, 108.7% w/w, 1H NMR (d6-DMSO) concordant with structure, no entrained solvent] as an off-white solid.
References:
ASTEX THERAPEUTICS LIMITED WO2008/9954, 2008, A1 Location in patent:Page/Page column 83-84; 105

5334-40-7
210 suppliers
$14.14/5gm:

138786-86-4
103 suppliers
$7.00/250mg

67-56-1
740 suppliers
$7.29/5ml-f

5334-40-7
210 suppliers
$14.14/5gm:

138786-86-4
103 suppliers
$7.00/250mg
75-36-5
559 suppliers
$17.92/100G

5334-40-7
210 suppliers
$14.14/5gm:

138786-86-4
103 suppliers
$7.00/250mg