(2E)-N-methoxy-3-(2-methoxyphenyl)-N-methylprop-2-enamide synthesis
- Product Name:(2E)-N-methoxy-3-(2-methoxyphenyl)-N-methylprop-2-enamide
- CAS Number:1422176-88-2
- Molecular formula:C12H15NO3
- Molecular Weight:221.25
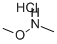
124931-12-0
88 suppliers
$21.00/250mg
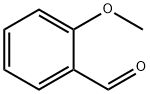
135-02-4
382 suppliers
$10.00/10g

1422176-88-2
3 suppliers
inquiry
Yield:1422176-88-2 100%
Reaction Conditions:
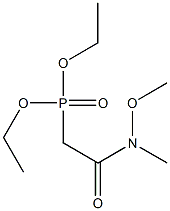
Stage #1: diethyl (N-methoxy-N-methylcarbamoylmethyl)phosphonatewith sodium hydride in tetrahydrofuran at 20;Horner-Wadsworth-Emmons Olefination;
Stage #2: ortho-anisaldehyde in tetrahydrofuran at -78 - 20;Horner-Wadsworth-Emmons Olefination;
Steps:
HWE Reaction; General Procedure A
General procedure: Diethyl 2-[methoxy(methyl)amino]-2-oxoethylphosphonate (1; 1.2 equiv) was dissolved in anhyd THF and NaH (1.2 equiv) was added portionwise at r.t. until gas evolution ceased. The mixture was then cooled to -78 °C and the appropriate benzaldehyde derivative (1 equiv) dissolved in anhyd THF was added. The reaction mixture was stirred at -78 °C for 1 h, then allowed to warm to r.t. and stirred overnight. H2O (10 mL) was added, the layers were separated, and the organic layer was washed with sat. aq NaHCO3 (2 × 10 mL). The combined aqueous phases were extracted with EtOAc (3 × 15 mL). The combined organic phases were then washed with brine (2 × 10 mL) ,dried (MgSO4), and concentrated in vacuo. If necessary, the crude product was purified by flash column chromatography (FCC) using the indicated eluent.
References:
Glas, Carina;Bracher, Franz [Synthesis,2019,vol. 51,# 3,p. 757 - 768]

98288-15-4
16 suppliers
$100.00/0.500g

1422176-88-2
3 suppliers
inquiry

135-02-4
382 suppliers
$10.00/10g

1422176-88-2
3 suppliers
inquiry