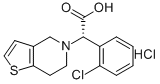
CLOPIDOGREL RELATED COMPOUND A (20 MG) ((S)-(O-CHLOROPHENYL)-6,7-DIHYDROTHIENO[3,2-C]PYRI-DINE-5(4H)-ACETIC ACID, HYDROCHLORIDE) synthesis
- Product Name:CLOPIDOGREL RELATED COMPOUND A (20 MG) ((S)-(O-CHLOROPHENYL)-6,7-DIHYDROTHIENO[3,2-C]PYRI-DINE-5(4H)-ACETIC ACID, HYDROCHLORIDE)
- CAS Number:144750-42-5
- Molecular formula:C15H14ClNO2S.ClH
- Molecular Weight:344.26
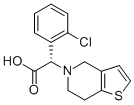
144457-28-3
97 suppliers
inquiry
![CLOPIDOGREL RELATED COMPOUND A (20 MG) ((S)-(O-CHLOROPHENYL)-6,7-DIHYDROTHIENO[3,2-C]PYRI-DINE-5(4H)-ACETIC ACID, HYDROCHLORIDE)](/CAS/GIF/144750-42-5.gif)
144750-42-5
122 suppliers
$39.00/500μg
Yield: 90%
Reaction Conditions:
with hydrogenchloride in methanol;water;acetone at 5 - 40; for 4 h;
Steps:
6 Preparation of (S)-(+)-α-(2-chlorophenyl)-6,7-dihydro-4H-thieno[3,2-c]pyridine-5-acetic acid hydrochloride (hydrochloride of formula (III))
EXAMPLE 6 Preparation of (S)-(+)-α-(2-chlorophenyl)-6,7-dihydro-4H-thieno[3,2-c]pyridine-5-acetic acid hydrochloride (hydrochloride of formula (III)) 61 g of (S)-(+)-α-(2-chlorophenyl)-6,7-dihydro-4H-thieno[3,2-c]pyridine-5-acetic acid obtained in Example 5 was added to a mixture of 300 ml of acetone and 15 ml of methanol, and then heated to 40° C. 16.4 ml of concentrated hydrochloric acid was added thereto, and the resulting mixture was stirred at room temperature for 2 hrs, and then, at below 5° C. for at least 2 hrs. The precipitated crystals were filtered, washed with 100 ml of acetone and dried at 40° C. under a reduced pressure, to obtain 61 g of the title compound as a white-yellow solid (yield: 90%). m.p.: 200-201° C. optical rotation: [α]D20+64.87° (c=1%, MeOH) optical purity: 99.5% ee (HPLC) 1H-NMR (DMSO-d6, ppm): δ 3.10 (s, 3H), 3.47 (s, 2H), 4.30 (s, 2H), 5.54 (s, 1H), 6.91 (d, 1H, J=4.8 Hz), 7.44 (d, 1H, J=4.8 Hz), 7.46-7.58 (m, 2H), 7.65 (d, 1H, 7.4 Hz), 7.97 (d, 1H, J=7.1 Hz) 13C-NMR (DMSO-d6, ppm): δ 21.78, 48.81, 50.17, 65.43, 125.04, 125.54, 127.99, 128.29, 128.36, 130.37, 130.46, 131.49, 132.02, 134.43, 167.53 IR (KBr, cm-1): 3435, 3114, 1728
References:
Hanmi Pharm., Co., Ltd. US2008/214821, 2008, A1 Location in patent:Page/Page column 5
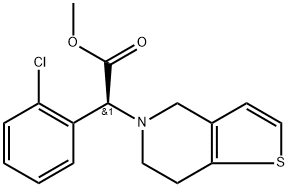
113665-84-2
294 suppliers
inquiry
![CLOPIDOGREL RELATED COMPOUND A (20 MG) ((S)-(O-CHLOROPHENYL)-6,7-DIHYDROTHIENO[3,2-C]PYRI-DINE-5(4H)-ACETIC ACID, HYDROCHLORIDE)](/CAS/GIF/144750-42-5.gif)
144750-42-5
122 suppliers
$39.00/500μg