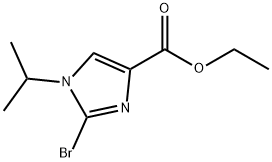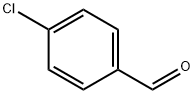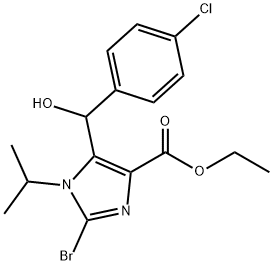
2-bromo-5-[(4-chlorophenyl)-hydroxy-methyl]-1-isopropyl-1H-imidazole-4-carboxylic acid ethyl ester synthesis
- Product Name:2-bromo-5-[(4-chlorophenyl)-hydroxy-methyl]-1-isopropyl-1H-imidazole-4-carboxylic acid ethyl ester
- CAS Number:1448869-68-8
- Molecular formula:C16H18BrClN2O3
- Molecular Weight:401.68
Yield:1448869-68-8 81%
Reaction Conditions:
Stage #1: 2-bromo-1-isopropyl-1H-imidazole-4-carboxylic acid ethyl esterwith lithium diisopropyl amide in tetrahydrofuran at -78; for 1.41667 h;Inert atmosphere;
Stage #2: 4-chlorobenzaldehyde in tetrahydrofuran at -78 - -20; for 0.666667 h;
Steps:
9.6 Step 9.6: ethyl 2-bromo-5-((4-chlorophenyl)(hydroxy) methyl)- 1 -isopropyl- 1 H-imidazole-4- carboxvlate
Ethyl 2-bromo-1-isopropyl-1H-imidazole-4-carboxylate (Step 9.5) (10 g, 38.3 mmol) was dissolved in THF (200 mL) under Ar and cooled to -78 00 Then 2M LDA (26.8 mL, 53.6 mmol) was added dropwise during 10 mm. The reaction mixture was stirred at -78 00 for 1 hr. 4- Chlorobenzaldehyde (7.3 g, 50 mmol), dissoved in THF (50 mL), was added during 10 mm. The reaction was stirred for 30 mm at -78 00 and then warmed up to -20 00 10 % aq. NH4CI solution (250 mL) was added dropwise. The resulting mixture was extracted three times with EtOAc. Combined extracts were washed with brine, dried over Na2504, filtered and concentrated under reduced pressure. Crystallization from iPr2O (100 mL) afforded the title product (12.5 g, 31.1 mmol, 81 % yield). tR: 1.16 mm (LC-MS 2); ESl-MS: 401/403 [M+H] (LC-MS 2).
References:
WO2014/191894,2014,A1 Location in patent:Page/Page column 74
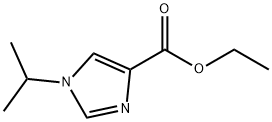
1260793-84-7
28 suppliers
inquiry
![2-bromo-5-[(4-chlorophenyl)-hydroxy-methyl]-1-isopropyl-1H-imidazole-4-carboxylic acid ethyl ester](/CAS/20180703/GIF/1448869-68-8.gif)
1448869-68-8
8 suppliers
inquiry
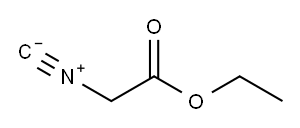
2999-46-4
216 suppliers
$13.43/1gm:
![2-bromo-5-[(4-chlorophenyl)-hydroxy-methyl]-1-isopropyl-1H-imidazole-4-carboxylic acid ethyl ester](/CAS/20180703/GIF/1448869-68-8.gif)
1448869-68-8
8 suppliers
inquiry

87951-12-0
0 suppliers
inquiry
![2-bromo-5-[(4-chlorophenyl)-hydroxy-methyl]-1-isopropyl-1H-imidazole-4-carboxylic acid ethyl ester](/CAS/20180703/GIF/1448869-68-8.gif)
1448869-68-8
8 suppliers
inquiry