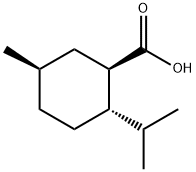
[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid synthesis
- Product Name:[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid
- CAS Number:16052-40-7
- Molecular formula:C11H20O2
- Molecular Weight:184.28
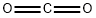
124-38-9
121 suppliers
$214.00/14L
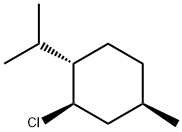
16052-42-9
72 suppliers
$45.00/250mg
![[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid](/CAS/GIF/16052-40-7.gif)
16052-40-7
56 suppliers
$40.00/1g
Yield:16052-40-7 93%
Reaction Conditions:
Stage #1: (1R,2S,5R)-menthyl chloridewith iodine;magnesium;ethylene dibromide at 70 - 75; for 4 h;Inert atmosphere;Autoclave;
Stage #2: carbon dioxide at 85 - 90; for 7.5 h;
Stage #3: with hydrogenchloride in water;Temperature;
Steps:
1 Example 1
The dried four-necked flask was added 9.4 g of magnesium juice,100 g of anhydrous dipropyl ether,And 0.1 g of iodine and 1 mL of 1,2-dibromoethane,Constant pressure dropping funnel by adding 60g of chlorinated menthane,150g anhydrous dipropyl ether and mixed evenly into the nitrogen protection;After the bottom material was heated to 70-75 ° C, the mixture of L-chlorobutane and dipropyl ether was added dropwise to the autoclave for 4 hours. After all the material is added, the kettle temperature rises to 85-90 ° C after 30 minutes of incubation.The dried CO2 gas was introduced into the prepared Grignard reagent with an amount of 0.15 eq / h and the reaction was terminated after 7 h.After the completion of the reaction, the Grignard material was slowly added dropwise to 190 g of a 6% HCl solution and quenched to pH = 1-2 and 30-35 ° C.The organic phase was recrystallized from 120 g of dipropyl ether under reduced pressure at -0.08 Mp to obtain levorotic acid. Yield 93%, content greater than 97%, optical rotation -45 ° (25 ° C ethanol). The dibutyl ether in the mother liquor after recrystallization was recovered under reduced pressure at -0.08 MPa, and the total solvent recovery was 96%.
References:
CN106431899,2017,A Location in patent:Paragraph 0084; 0085; 0086; 0087; 0088; 0089; 0090-0104
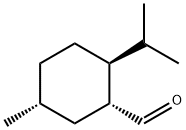
80434-59-9
3 suppliers
inquiry
![[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid](/CAS/GIF/16052-40-7.gif)
16052-40-7
56 suppliers
$40.00/1g
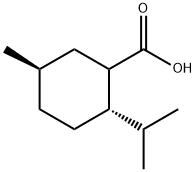
663218-94-8
0 suppliers
inquiry
![[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid](/CAS/GIF/16052-40-7.gif)
16052-40-7
56 suppliers
$40.00/1g
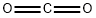
124-38-9
121 suppliers
$214.00/14L

16052-42-9
72 suppliers
$45.00/250mg
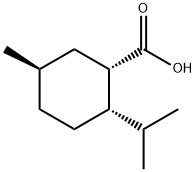
70985-58-9
1 suppliers
inquiry
![[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid](/CAS/GIF/16052-40-7.gif)
16052-40-7
56 suppliers
$40.00/1g

2216-51-5
664 suppliers
$5.00/100mg
![[1R-(1alpha,2beta,5alpha)]-2-(isopropyl)-5-methylcyclohexanecarboxylic acid](/CAS/GIF/16052-40-7.gif)
16052-40-7
56 suppliers
$40.00/1g