
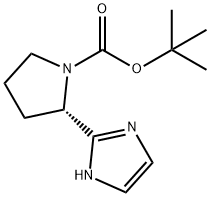
1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)- synthesis
- Product Name:1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)-
- CAS Number:1613055-24-5
- Molecular formula:C18H30BN3O4
- Molecular Weight:363.26

1228552-62-2
10 suppliers
inquiry
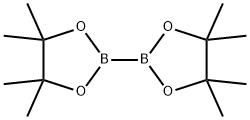
73183-34-3
566 suppliers
$6.00/5g
![1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)-](/CAS/20210111/GIF/1613055-24-5.gif)
1613055-24-5
3 suppliers
inquiry
Yield:1613055-24-5 85%
Reaction Conditions:
with dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2;potassium acetate in N,N-dimethyl-formamide at 90; for 6 h;Inert atmosphere;Sealed tube;
Steps:
5.5 the preparation of compound 5-6
A mixture of compound 1-20 (0.36 g, 1.0 mmol), compound 1-14-2 (0.30 g, 1.2 mmol), Pd(dppf)Cl2'CH2Cl2 (40.8 mg, 0.05 mmol) and KOAc (0.25 g, 2.5 mmol) in DMF (5.0 mL) was stirred at 90 °C under N2 for 6.0 hrs. After the reaction was completed, the mixture was cooled to rt, diluted with EtOAc (50 mL) and filtered through a celite pad. The filtrate was washed with water (30 mL x 3) and brine, dried over anhydrous Na2S04 and concentrated in vacuo. The residue was purified by silica gel column chromatography (PE/EtOAc (v/v) = 1/2) to give the title compound (0.31 g, 85%). The compound was characterized by the following spectroscopic data: MS (ESI, pos.ion) mlz: 364.5 [M+H]+; and NMR (400 MHz, CDC13) δ (ppm): 7.50 (s, 1H), 4.93-4.88 (m, 1H), 3.59-3.53 (m, 1H), 3.28-3.21 (m, 1H), 2.38-2.30 (m, 1H), 2.27-2.12 (m, 2H), 2.08-1.98 (m, 1H), 1.41 (s, 9H), 1.40-1.39 (m, 6H), 1.37-1.36 (m, 6H).
References:
WO2014/82380,2014,A1 Location in patent:Paragraph 00371; 00376
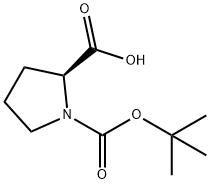
15761-39-4
603 suppliers
$5.00/10g
![1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)-](/CAS/20210111/GIF/1613055-24-5.gif)
1613055-24-5
3 suppliers
inquiry
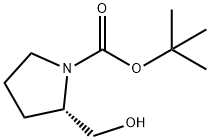
69610-40-8
383 suppliers
$6.00/5g
![1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)-](/CAS/20210111/GIF/1613055-24-5.gif)
1613055-24-5
3 suppliers
inquiry
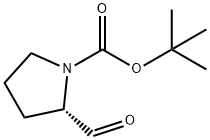
69610-41-9
250 suppliers
$9.00/1g
![1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)-](/CAS/20210111/GIF/1613055-24-5.gif)
1613055-24-5
3 suppliers
inquiry
1007882-58-7
58 suppliers
$65.00/100mg
![1-Pyrrolidinecarboxylic acid, 2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-imidazol-2-yl]-, 1,1-dimethylethyl ester, (2S)-](/CAS/20210111/GIF/1613055-24-5.gif)
1613055-24-5
3 suppliers
inquiry