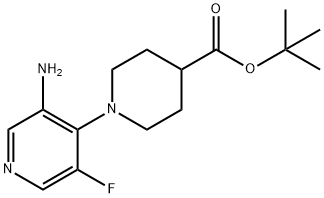
Tert-Butyl 1-(3-Amino-5-Fluoropyridin-4-Yl)Piperidine-4-Carboxylate synthesis
- Product Name:Tert-Butyl 1-(3-Amino-5-Fluoropyridin-4-Yl)Piperidine-4-Carboxylate
- CAS Number:1613192-01-0
- Molecular formula:C15H22FN3O2
- Molecular Weight:295.35
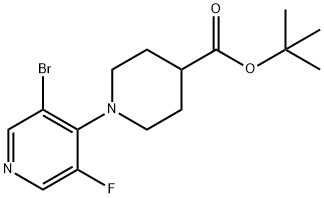
1613193-36-4
12 suppliers
inquiry

1613192-01-0
16 suppliers
$233.59/100mg
Yield:1613192-01-0 86.5%
Reaction Conditions:
Stage #1: tert-butyl 1-(3-bromo-5-fluoro-4-pyridyl)piperidine-4-carboxylatewith Benzophenone imine;tris-(dibenzylideneacetone)dipalladium(0);caesium carbonate;4,5-bis(diphenylphosphino)-9,9-dimethylxanthene in 1,4-dioxane at 95;Inert atmosphere;
Stage #2: with hydrogenchloride in 2-methyltetrahydrofuran;water at 20; for 0.5 h;Solvent;
Steps:
N-15.2; N-15.3 Step 2: tert-butyl 1-(3-amino-5-fluoro-4-pyridyl)piperidine-4-carboxylate 27
To a solution of tert-butyl l-(3-bromo-5-fluoro-4-pyridyl)piperidine-4-carboxylate 26 (98 g, 272.8 mmol), diphenylmethanimine (59.34 g, 54.94 mL, 327.4 mmol) and Cs2C03 (177.8 g, 545.6 mmol) in Dioxane (1.274 L) was added xantphos (15.78 g, 27.28 mmol) and Pd2(dba)3 (12.49 g, 13.64 mmol). The mixture was stirred under nitrogen at 95°C overnight. The mixture was cooled to room temperature then partitioned between EtO Ac (1000 mL, 10 vol eq.) and water (490 mL, 5 vol eq.), mixed and organic layer separated. Organics washed further with water (1 x 250 mL), brine (250 mL), dried (MgS04), filtered and concentrated in vacuo to leave crude product as a dark red viscous oil, 185.3g. [00381] The obtained product oil (185.3g) was dissolved in THF (882.0 mL) and HCl (545.5 mL of 2 M, 1.091 mol) was added. The resulting mixture was stirred at room temperature for 20 mins. THF was removed in vacuo then additional (HCl (2M) (588.0 mL) was added. The aqueous was washed twice with EtO Ac (294.0 mL). A large amount of a yellow precipitate formed during extraction in both organic and aqueous phase, the solid from both the organic and aqueous phase was collected by filtration and dried by suction. The mixed organic and aqueous filtrate was added to separating funnel, extracted with 2M HCl (2 x 200 mL). All aqueous phases plus solid collected on sinter (product) were combined to give a suspension. The pH was adjusted to 6 using 2M NaOH and extracted with DCM (3 x 600 mL). The organics were combined, dried (MgS04), filtered and concentrated in vacuo to leave a pale orange waxy solid, 1 12.2g. This solid was slurried in MeCN (200 mL), stirred for 10 mins then solid collected by filtration, washed with minimal MeCN and dried by suction to leave product 27 as a white solid (66.8 g, 83% yield). XH NMR (500 MHz, DMSO- d6) δ 7.82 (d, 1H), 7.63 (d, 1H), 5.22 (s, 2H), 3.1 1 - 3.00 (m, 2H), 2.91 (tt, 2H), 2.36 (tt, 1H), 1.88 - 1.83 (m, 2H), 1.79 - 1.71 (m, 2H), 1.43 (s, 9H). MS (ES+) 297.1.; Tert-butyl l-(3-bromo-5-fluoropyridin-4-yl)piperidine-4-carboxylate 26 (800g, 2.23mol) was dissolved in 1,4-dioxane (7.5L). Diphenylmethanimine (484g, 2.67mol) was added in one portion followed by cesium carbonate (1.45Kg, 4.45mol), xantphos (129g, 223mmol) and Pd2(dba)3 (102g, 1 1 lmmol). Additional 1,4-dioxane (2.9L) was added and the mixture heated to 95°C under nitrogen until the reaction was complete (determined by HPLC analysis). The mixture was cooled to 20°C and ethyl acetate (8L) and water (4L) were added. The organic phase was isolated and washed with water (4L) and brine (3.5L) and dried over magnesium sulphate and filtered. The filtrate was concentrated to a brown oil (1.3Kg). The oil was dissolved in 2-methyltetrahydrofuran (7.2L) and 2M HC1 was added at 20°C and the mixture stirred for 30 minutes. The aqueous layer was isolated and the organic layer extracted with 2M HC1 (1.2L). The combined aqueous was neutralised with 2M NaOH (5.4L, pH 8-9). The product was extracted into 2-methyltetrahydrofuran (14L then 2x5L). The combined extracts were washed with water (1.6L) and the organic solution concentrated. The residue was slurried in acetonitrile (2L), filtered and dried. This gave the product 27 as a white solid (568.7g, 86.5%); 1H MR (500 MHz, DMSO-i) δ 7.82 (d, 1H), 7.63 (d, 1H), 5.22 (s, 2H), 3.11 - 3.00 (m, 2H), 2.91 (tt, 2H), 2.36 (tt, 1H), 1.88 - 1.83 (m, 2H), 1.79 - 1.71 (m, 2H), 1.43 (s, 9H); 19F NMR (500 MHz, DMSO-d6) δ - 140.0; MS (ES+) 297.1.
References:
WO2014/89379,2014,A1 Location in patent:Paragraph 00380; 00381; 00385
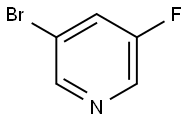
407-20-5
345 suppliers
$10.00/1g

1613192-01-0
16 suppliers
$233.59/100mg

1613192-96-3
13 suppliers
inquiry

1613192-01-0
16 suppliers
$233.59/100mg
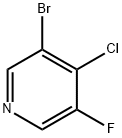
1211540-92-9
77 suppliers
$45.00/50mg

1613192-01-0
16 suppliers
$233.59/100mg
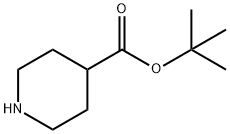
138007-24-6
74 suppliers
$65.00/50mg

1613192-01-0
16 suppliers
$233.59/100mg