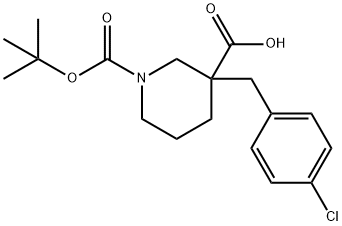
1-[(TERT-BUTYL)OXYCARBONYL]-3-(4-CHLOROBENZYL)PIPERIDINE-3-CARBOXYLIC ACID synthesis
- Product Name:1-[(TERT-BUTYL)OXYCARBONYL]-3-(4-CHLOROBENZYL)PIPERIDINE-3-CARBOXYLIC ACID
- CAS Number:174606-16-7
- Molecular formula:C18H24ClNO4
- Molecular Weight:353.84
![1-BOC-3-[(4-CHLOROPHENYL)METHYL]-3-PIPERIDINECARBOXYLIC ACID ETHYL ESTER](/CAS/GIF/174606-15-6.gif)
174606-15-6
0 suppliers
inquiry
![1-[(TERT-BUTYL)OXYCARBONYL]-3-(4-CHLOROBENZYL)PIPERIDINE-3-CARBOXYLIC ACID](/CAS/GIF/174606-16-7.gif)
174606-16-7
35 suppliers
$52.00/50 mg
Yield:-
Reaction Conditions:
with hydrogenchloride;sodium hydroxide
Steps:
2.B N-boc-3-(4-chlorobenzyl)piperidin-3-carboxylic acid
Step B N-boc-3-(4-chlorobenzyl)piperidin-3-carboxylic acid A suspension of Ethyl-3-(4-chlorobenzyl)-N-Boc-piperidin-3-carboxylate (36.4 g, 0.105 mol) in isopropyl alcohol (300 mL) was treated with 10 N NaOH (300 mL). The mixture was heated at reflux for 48 hours. The reaction was cooled to room temperature and carefully neutralized by pouting over 1 L of crushed ice and adding 6 N HCl until pH 3. The mixture was then extracted with ethyl acetate (3*500 mL). The combined organic extracts were dried over anhydrous magnesium sulfate, filtered, and concentrated at reduced pressure to give 31 g of the product. 1 H NMR CDCl3 δ10.18 (br s), 7.26 (d, J=8.5 Hz, 2H), 7.04 (d, J=8.5 Hz, 2H), 3.95 (m, 1H), 3.6 (m, 1H), 3.2 (m, 2H), 2.90 (d, J=12 Hz, 1H), 2.78 (d, J =12 Hz, 1H), 2.00 (m, 1H), 1.75-1.30 (m, 3H), 1.44 (s, 9H).
References:
Merck & Co., Inc. US5475109, 1995, A