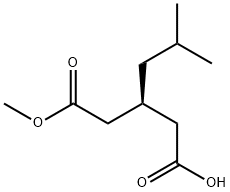
(3S)-3-(2-methoxy-2-oxoethyl)-5-methylhexanoic acid synthesis
- Product Name:(3S)-3-(2-methoxy-2-oxoethyl)-5-methylhexanoic acid
- CAS Number:181289-25-8
- Molecular formula:C10H18O4
- Molecular Weight:202.25
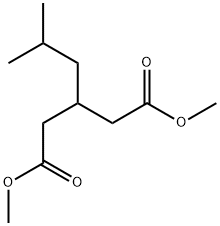
145328-03-6
24 suppliers
inquiry

181289-25-8
13 suppliers
inquiry
Yield:181289-25-8 96%
Reaction Conditions:
with water;sodium hydroxide;lipase B from Candida antarctica in tert-Amyl alcohol at -2; pH=7.3; for 96 h;Product distribution / selectivity;Aqueous phosphate buffer;Enzymatic reaction;
Steps:
7
Example 7: Preparation of S-IBG-Me-ester (S-iso-butyl glutaric methyl ester)[0062] IBG-dimethyl ester (10.6 g) was suspended in 0.05 M potassium phosphate buffer (60 ml) and tert-pentanol (10 ml) in a jacketed reactor equipped with pH probe and magnetically stirred. The mixture was cooled to -2°C and CaL-B liquid from Candida Antartica (1 ml, Novozymes; 7000 TBU/ml) was added. The reaction was stirred for 96 h, the pH of the reaction was kept constant at 7.3 by addition of 2 M NaOH (pH-stat). The mixture was extracted with MTBE (2 x 10 ml), the organic phase was extracted with NaHCO3 (10 ml). The water-phase was acidified with cone. HCl to pH 1.5 and extracted with MTBE (3 x 10ml). Evaporation of the combined extracts yielded 9.5 g colorless oil of S-IBG-Me-ester. 95.5% optical pure (96% yield).
References:
WO2009/158343,2009,A1 Location in patent:Page/Page column 15

67-56-1
738 suppliers
$9.00/25ml
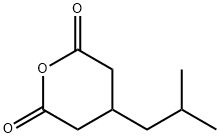
185815-59-2
116 suppliers
inquiry

181289-25-8
13 suppliers
inquiry
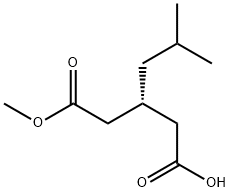
156048-92-9
2 suppliers
inquiry
