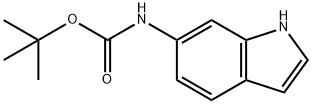
(1H-INDOL-6-YL)-CARBAMIC ACID TERT-BUTYL ESTER synthesis
- Product Name:(1H-INDOL-6-YL)-CARBAMIC ACID TERT-BUTYL ESTER
- CAS Number:885273-73-4
- Molecular formula:C13H16N2O2
- Molecular Weight:232.28
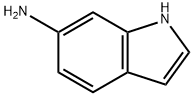
5318-27-4
229 suppliers
$15.00/250mg

24424-99-5
824 suppliers
$13.50/25G

885273-73-4
24 suppliers
inquiry
Yield:885273-73-4 97%
Reaction Conditions:
with sodium hydrogencarbonate in tetrahydrofuran;water at 20; for 18 h;
Steps:
31 Synthesis of tert-Butyl 1H-indol-6-ylcarbamate (compound 64-2)
Synthesis of tert-Butyl 1H-indol-6-ylcarbamate (compound 64-2)[0000222] To a stirred solution of 1H-indol-6-amine (1.12 g, 8.5 mmol, 1 equiv) was added di-tert-butyl dicarbonate (1.95 g, 8.9 mmol, 1.05 equiv) in tetrahydrofuran (6 mL) . Saturated aqueous sodium bicarbonate (6 mL) was added and the reaction was stirred at room temperature for 18 hours. The layers were separated and the aqueous layer was extracted with ethyl acetate (3 x 5 mL) . The organic layers were combined, dried over sodium sulfate and concentrated under reduced pressure to give compound 64-2 as a white solid (2.2 g, 97% yield)
References:
WO2013/123215,2013,A2 Location in patent:Paragraph 0000222

19727-83-4
200 suppliers
$11.00/1g

885273-73-4
24 suppliers
inquiry
4769-96-4
263 suppliers
$9.00/250mg

885273-73-4
24 suppliers
inquiry
496-15-1
422 suppliers
$8.00/10g

885273-73-4
24 suppliers
inquiry