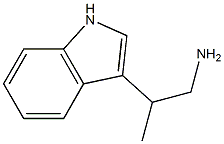
2-(1H-indol-3-yl)propan-1-amine synthesis
- Product Name:2-(1H-indol-3-yl)propan-1-amine
- CAS Number:4765-22-4
- Molecular formula:C11H14N2
- Molecular Weight:174.2423
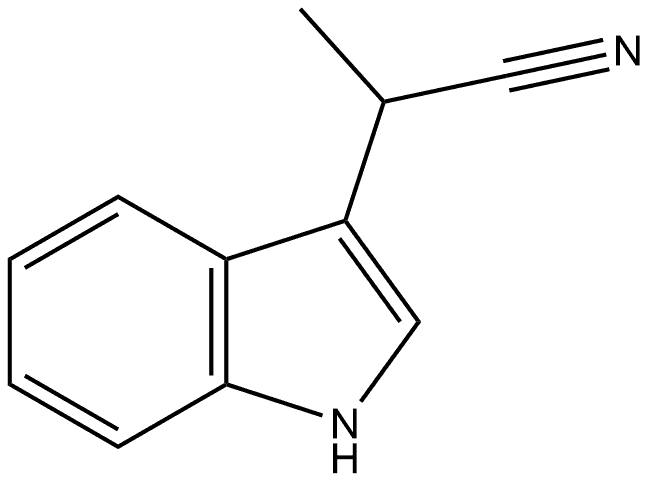
20862-85-5
0 suppliers
inquiry

4765-22-4
6 suppliers
inquiry
Yield:4765-22-4 82%
Reaction Conditions:
with lithium aluminium tetrahydride in tetrahydrofuran;
Steps:
32.D PREPARATION OF ETHYL 3-(4-FLUOROBENZOYL)-1-METHYL-1,2,3,6-TETRAHYDROAZEPINO[4,5-B]INDOLE-5-CARBOXYLATE
D. To a stirred suspension of LAH (3.4 g, 90 mmol) in THF (40 mL, anhyd) chilled to 0° C. was added a solution of 2-(1H-indol-3-yl)propionitrile (2.5 g, 14.7 mmol) in THF (40 mL, anhyd). The reaction mixture was allowed to warm to ambient temperature and then heated at reflux. After 1 hour, the reaction mixture was cooled to 0° C. and quenched cautiously with wet THF (5-10% H2O) until gas evolution had ceased. The resulting mixture was filtered through Celite and concentrated to give a brown residue. The filtering agent was rinsed with Et2O (100 mL), which was combined with the residue, dried (Na2SO4), and concentrated under reduced pressure to yield 2-(1H-indol-3-yl)propylamine (2.1 g, 82%) as a pale amber oil; 1H-NMR (CDCl3): δ 8.28 (1H, br s), 7.61 (1H, d), 7.36 (1H, d), 7.16 (1H, app t), 7.08 (1H, app t), 6.96 (1H, br s), 3.41 (2H, br s), 3.19 (1H, q), 2.95 (2H, app d), 1.35 (3H, d).
References:
US2005/54634,2005,A1
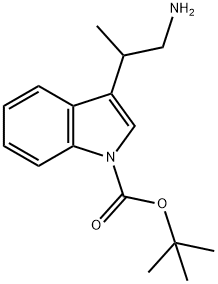
783349-72-4
0 suppliers
inquiry

4765-22-4
6 suppliers
inquiry
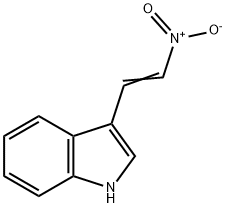
3156-51-2
31 suppliers
$28.60/50mg

4765-22-4
6 suppliers
inquiry
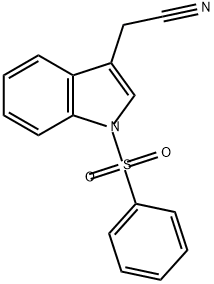
143703-45-1
0 suppliers
inquiry

4765-22-4
6 suppliers
inquiry
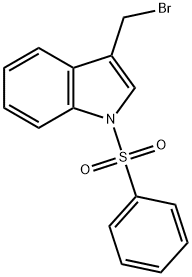
58550-78-0
0 suppliers
inquiry

4765-22-4
6 suppliers
inquiry