
2-(2-METHYL-1H-INDOL-1-YL)ETHANAMINE synthesis
- Product Name:2-(2-METHYL-1H-INDOL-1-YL)ETHANAMINE
- CAS Number:883535-89-5
- Molecular formula:C11H14N2
- Molecular Weight:174.24
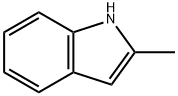
95-20-5
407 suppliers
$5.00/10g
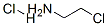
870-24-6
514 suppliers
$5.00/5g

883535-89-5
27 suppliers
$143.00/100mg
Yield:883535-89-5 99%
Reaction Conditions:
with tetra(n-butyl)ammonium hydrogensulfate;sodium hydroxide in toluene; for 17 h;Reflux;Inert atmosphere;
Steps:
A.1.1.1 A.1.1.1. 2-(2-Methyl-1 H-indol-1-yl)ethan-1-amine
To a solution of 2-methylindole (10.04 g, 75 mmol) in toluene (200 mL) are added 2-chloroethylamine hydrochloride (17.4 g, 150 mmol), freshly powdered NaOH (21.00 g, 525 mmol) and tetrabutyl ammonium hydrogen sulfate (2.037 g, 6 mmol). The resulting mixture is heated up to reflux and stirred for 17h. It is then cooled down to RT, and filtered through a filter paper. The residue is triturated twice with toluene, and filtrated. The filtrate is concentrated under reduced pressure, and the residue is purified by FC, using a gradient of DCM/MeOH from 100:0 to 95:5. After concentration of the product containing fractions, the title compound (13.2 g, 99%) is obtained as a yellow resin: LC-MS A: tR = 0.54 min; [M+H]+ = 175.31.
References:
WO2017/85198,2017,A1 Location in patent:Page/Page column 120
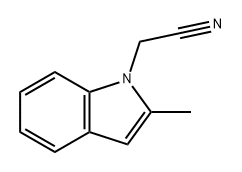
104894-21-5
0 suppliers
inquiry

883535-89-5
27 suppliers
$143.00/100mg

95-20-5
407 suppliers
$5.00/10g

883535-89-5
27 suppliers
$143.00/100mg