
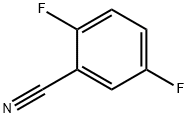
2,5-DIFLUOROTHIOBENZAMIDE synthesis
- Product Name:2,5-DIFLUOROTHIOBENZAMIDE
- CAS Number:1029623-72-0
- Molecular formula:C7H5F2NS
- Molecular Weight:173.18
64248-64-2
232 suppliers
$10.00/10g

1029623-72-0
11 suppliers
$61.89/1g
Yield:1029623-72-0 99%
Reaction Conditions:
with pyridine;ammonium sulfide;triethylamine in water at 50; for 5 h;
Steps:
1
Synthesis of 2,5-difluorobenzothioamide; A stirred solution of 2,5-difluorobenzonitrile (25 g, 180 mmol) in pyridine (90 mL) was treated with 20 wt % ammonium sulfide in water (67.4 mL, 198 mmol) and triethylamine (27.4 mL, 198 mmol). The reaction mixture was stirred at 50° C. for 5 hr until the reaction was complete. After cooling to room temperature, the mixture was diluted with cold water and extracted with EtOAc. The organic layer was separated, then washed with H2O (×3), brine (×3), then dried over anhydrous Na2SO4, filtered, and evaporated under reduced pressure to give the crude product. Purification on silica gel column (20% EtOAc in hexanes) to afford 2,5-difluorobenzothioamide as yellow solid (31.0 g, 99%). 1H NMR (CDCl3, 300 MHz): δ 7.12 (m, 2H), 7.90 (br, 2H), 8.08 (m, 1H). LC/MS (uplc): MH+174.0, 0.64 min.
References:
US2011/256128,2011,A1 Location in patent:Page/Page column 13; 23-24