
2-(BENZYLOXY)-6-BROMONAPHTHALENE synthesis
- Product Name:2-(BENZYLOXY)-6-BROMONAPHTHALENE
- CAS Number:2234-45-9
- Molecular formula:C17H13BrO
- Molecular Weight:313.19
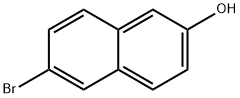
15231-91-1
393 suppliers
$7.00/5g
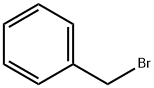
100-39-0
424 suppliers
$10.00/10g

2234-45-9
45 suppliers
$20.00/250mg
Yield:2234-45-9 95%
Reaction Conditions:
Stage #1: 6-bromo-naphthalen-2-olwith sodium hydride in N,N-dimethyl-formamide;mineral oil at 0;
Stage #2: benzyl bromide in N,N-dimethyl-formamide;mineral oil;
Steps:
1.6
In a dry single neck round bottom flask, sodium hydride (460 mg, 20 mmol, 2 equiv.) was added to 6-bromo- 2-naphthol (2.2 g, 10 mmol, 1 equiv.) in anhydrous ^.A'-dimethylformamide (15 mL) at 0 °C. Approximately 15-20 minutes later, benzyl bromide (1.2 mL, 10 mmol, 1 equiv.) was added drop- wise and the stirring was continued until the starting material was completely consumed. It takes generally 3-4 h for the reaction to go to completion. Saturated aqueous NaHC03 was added to the reaction mixture, and the solution was extracted with EtOAc. The combined organic layers were washed with water and saturated aqueous NaCl solution, dried over anhydrous Na2S04, filtered, and concentrated. The residue was purified on silca gel to provide the desired product with 95% of yield as white solid.
References:
WO2012/79154,2012,A1 Location in patent:Page/Page column 99

15231-91-1
393 suppliers
$7.00/5g
100-44-7
630 suppliers
$13.50/250G

2234-45-9
45 suppliers
$20.00/250mg