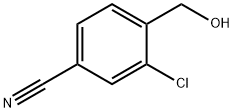
2-Chloro-4-cyanobenzyl alcohol synthesis
- Product Name:2-Chloro-4-cyanobenzyl alcohol
- CAS Number:1261618-25-0
- Molecular formula:C8H6ClNO
- Molecular Weight:167.59
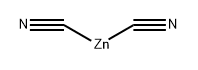
557-21-1
86 suppliers
$12.00/5g
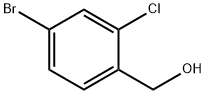
185315-48-4
57 suppliers
$55.00/1g

1261618-25-0
20 suppliers
inquiry
Yield:1261618-25-0 40%
Reaction Conditions:
with tetrakis(triphenylphosphine) palladium(0) in N,N-dimethyl-formamide at 100; for 16 h;Inert atmosphere;
Steps:
1.3 1.3 3-chloro-4-hydroxymethyl benzonitrile (1-2)
Zinc cyanide (0.67 g, 5.73 mmol) and tetrakis(triphenylphosphine) palladium (Pd(PPh3)4, 0.33 g, 0.287 mmol) were added into a solution of 4-bromo-2-chlorobenzyl alcohol (1-1, 1.27 g, 5.73 mmol) in DMF (15 mL). After deoxygenated via argon bubbling, the reaction mixture was heated at 100°C and reacted for 16 hours, cooled down to room temperature, diluted with ethyl acetate (50 mL), washed successively with water (50 mL*3) and saturated brine (50 mL*3), dried with anhydrous sodium sulfate and filtered. The filtrate was concentrated to obtain a crude product. The crude product was then purified by column chromatography (elution system: petroleum ether: ethyl acetate = 15/1-4/1) to obtain a white solid product (0.387 g, 40% yield). The molecular ion peak shown by liquid chromatography-mass spectrometry was: MS (ESI): m/z 168.0/170.1 [M+H]+.
References:
EP3048103,2016,A1 Location in patent:Paragraph 0067; 0068
59748-90-2
275 suppliers
$8.00/1g

1261618-25-0
20 suppliers
inquiry

185312-82-7
91 suppliers
$8.00/250mg

1261618-25-0
20 suppliers
inquiry