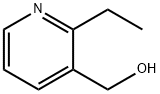
(2-ethylpyridin-3-yl)methanol synthesis
- Product Name:(2-ethylpyridin-3-yl)methanol
- CAS Number:15440-72-9
- Molecular formula:C8H11NO
- Molecular Weight:137.18
3421-76-9
64 suppliers
$60.00/100mg

15440-72-9
9 suppliers
inquiry
Yield:15440-72-9 250 mg
Reaction Conditions:
with lithium aluminium tetrahydride in tetrahydrofuran; for 1 h;Inert atmosphere;Cooling with ice;
Steps:
68
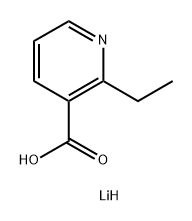
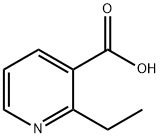
LiAlH4 (9.0 mL of a 1 M solution in THF, 9.0 mmol) was added dropwise to a suspension of 2-ethylpyridine-3-carboxylic acid (500 mg, 3.31 mmol) in THF (5 mL) under N2 with cooling in an ice bath. The resulting solution was stirred for 1 hour, then quenched by the careful addition of water (0.35 mL) then aqueous NaOH (0.25 mL of a 15% w/v solution) then water (1 mL). After stirring for 10 minutes the mixture was filtered and the filtrate concentrated in vacuo to give a colourless oil (250 mg); MS m/z: 138 (M+H)+. This material was dissolved in DCM (10 mL) and tert-butyl N-methylsulfonylcarbamate (630 mg, 3.23 mmol) and PPh3 (950 mg, 3.62 mmol) were added followed by DEAD (500 μL, 3.04 mmol). The reaction mixture was stirred at ambient temperature for 18 hours, then partitioned between EtOAc and saturated aqueous sodium bicarbonate solution. After stirring for 5 minutes the organic phase was isolated using a phase separation cartridge. The solvent was removed in vacuo and the residue purified by column chromatography (silica, DCM-EtOAc elution) to give an oil that was taken up in HCl (10 mL of a 3 M solution in MeOH). PtO2 (150 mg, 0.66 mmol) was added and the mixture shaken in a Parr hydrogenator for 18 hours under 60 psi of H2 pressure. The reaction mixture was poured onto a pre-wetted ion-exchange cartridge, washing with MeOH then eluting the product with 2 M methanolic NH3 solution. The filtrate was concentrated in vacuo to give N-((2-ethylpiperidin-3- yl)methyl)methanesulfonamide A74 as a colourless oil (70 mg) that was taken directly on to the next reaction without further purification; MS m/z: 221 (M+H)+.
References:
WO2019/148132,2019,A1 Location in patent:Paragraph 00707; 00708
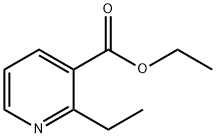
3197-39-5
4 suppliers
inquiry

15440-72-9
9 suppliers
inquiry

64114-26-7
5 suppliers
inquiry

15440-72-9
9 suppliers
inquiry