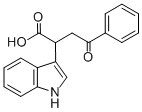
2-INDOL-3-YL-4-OXO-4-PHENYLBUTANOIC ACID synthesis
- Product Name:2-INDOL-3-YL-4-OXO-4-PHENYLBUTANOIC ACID
- CAS Number:6266-66-6
- Molecular formula:C18H15NO3
- Molecular Weight:293.32
120-72-9
624 suppliers
$6.00/25g

17812-07-6
52 suppliers
inquiry

6266-66-6
62 suppliers
$186.00/500mg
Yield: 75%
Reaction Conditions:
in benzene at 80; for 5 h;
Steps:
1 4-Phenyl-2-(1H-indol-3-yl)-4-oxo-butanoic acid (Compound No.20)
4-Phenyl-2-(1H-indol-3-yl)-4-oxo-butanoic acid (Compound No.20) In a 30-mL round-bottomed flask, trans-4-phenyl-4-oxo-2-butenoic acid (1.0 g, 5.65 mmol) was dissolved in benzene (25 mL). To the solution, indole (0.79 g, 6.77 mmol) was added, and the mixture was stirred at 80° C. for 5 hours and stirred until the temperature became room temperature. The solvent in the reaction solution was distilled off under reduced pressure, and the residue was recrystallized from benzene to obtain 4-phenyl-2-(1H-indol-3-yl)-4-oxo-butanoic acid (compound No.20) (1.24 g, yield: 75%); Melting point: 149 to 150° C.; 1H NMR (400 MHz, acetone-df): δ 10.17 (1H, brs, 1H), 8.05 (2H, d, J=8.2 Hz), 7.80 (1H, d, J=8.3 Hz), 7.57 (1H, t, J=7.8 Hz), 7.51 (2H, dd, J=8.2, 7.8 Hz), 7.41 (1H, d, J=8.2 Hz), 7.37 (1H, s), 7.13 (1H, t, J=8.2 Hz), 7.06 (1H, t, J=8.2 Hz), 4.57 (1H, dd, J=11.0, 4.1 Hz), 4.13 (1H, dd, J=17.8, 11.0 Hz), 3.41 (1H, dd, J=17.8, 4.1 Hz); IR: (neat): 3400, 3055, 1711, 1677, 1453 cm-1; FAB-MS m/z [M+H]+ calcd for 294.1130 (C18H16NO3), found 294.1143 [M+H]+.
References:
TOHOKU UNIVERSITY;KAKE EDUCATIONAL INSTITUTION;KANAGAWA PREFECTURAL HOSPITAL ORGANIZATION KANAGAWA CHILRDEN'S MEDICAL CENTER;ABE, Takaaki;TOMINAGA, Teiji;HAYASHI, Kenichiro;OSAKA, Hitoshi US2015/353489, 2015, A1 Location in patent:Paragraph 0207; 0208