
2-Methylsulfanyl-7,8-dihydro-5H-pyrido[4,3-d]pyriMidine -6-carboxylic acid tert-butyl ester synthesis
- Product Name:2-Methylsulfanyl-7,8-dihydro-5H-pyrido[4,3-d]pyriMidine -6-carboxylic acid tert-butyl ester
- CAS Number:259809-78-4
- Molecular formula:C13H19N3O2S
- Molecular Weight:281.37
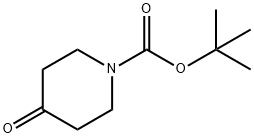
![1-Boc-3-[(Dimethylamino)methylene]-4-oxopiperidine](/CAS/GIF/157327-41-8.gif)
157327-41-8
53 suppliers
$24.00/1g
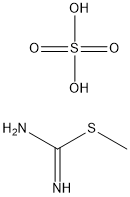
867-44-7
369 suppliers
$5.00/10g
![2-Methylsulfanyl-7,8-dihydro-5H-pyrido[4,3-d]pyriMidine
-6-carboxylic acid tert-butyl ester](/CAS/GIF/259809-78-4.gif)
259809-78-4
26 suppliers
inquiry
Yield:259809-78-4 37%
Reaction Conditions:
with sodium hydroxide in water at 75; for 2.5 h;
Steps:
22 t-Butyl-2-(methylthio)-5,6,7,8-tetrahydropyrido[4,3-d]pyrimidine-6-carboxylate
Preparation 22
t-Butyl-2-(methylthio)-5,6,7,8-tetrahydropyrido[4,3-d]pyrimidine-6-carboxylate
A suspension of t-Butyl-3-[(dimethylamino)methylidene]-4-oxopiperidine-1-carboxylate (10.95 g, 43.05 mmol) and 2-methyl-2-thiopseudourea hemisulfate (7.79 g, 55.97 mmol) in water (175 ml) was treated with 1.0N NaOH(aq.) (46.63 ml, 46.63 mmol) and the reaction mixture was heated at 75° C. for 2.5 hours.
The aqueous mixture was extracted with dichloromethane and the organic extract was dried (MgSO4), filtered, and concentrated.
Elution through a flash column (silica gel 60, 230-400 mesh, 7:3 hexanes:EtOAc) gave the title compound as a yellow oil (4.45 g, 37%).
References:
US2013/237538,2013,A1 Location in patent:Paragraph 0254
79099-07-3
543 suppliers
$5.00/5g
![2-Methylsulfanyl-7,8-dihydro-5H-pyrido[4,3-d]pyriMidine
-6-carboxylic acid tert-butyl ester](/CAS/GIF/259809-78-4.gif)
259809-78-4
26 suppliers
inquiry