
tert-Butyl (1R*,4R*)-4-(benzylamino)cyclohexylcarbamate synthesis
- Product Name:tert-Butyl (1R*,4R*)-4-(benzylamino)cyclohexylcarbamate
- CAS Number:264608-33-5
- Molecular formula:C18H28N2O2
- Molecular Weight:304.43
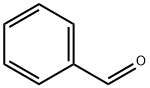
100-52-7
944 suppliers
$20.37/250g

177906-48-8
213 suppliers
$6.00/250mg

264608-33-5
7 suppliers
inquiry
Yield:264608-33-5 69 %
Reaction Conditions:
Stage #1: benzaldehyde;trans-N-Boc-1,4-cyclohexanediaminewith acetic acid in methanol at 20;
Stage #2: with methanol;sodium tetrahydroborate at 20;
Steps:
Step 1. tert-Butyl ((1r,4r)-4-(benzylamino)cyclohexyl)carbamate.
To a solution of tert-butyl ((1r,4r)-4-aminocyclohexyl)carbamate (0.5 g, 2.33 mmol) in MeOH (15 mL), benzaldehyde (1.2 mL, 11.67 mmol) and AcOH (0.13 mL, 2.33 mmol) were added and the solution was stirred at rt overnight. Then, a mixture of NaBH4(0.88 g, 23.3 mmol) in MeOH (10 mL) was added and the reaction mixture was stirred at rt for 1 h. Then, it was cooled to 0 C and 10 wt% NaOH aq. sol. (10 mL) was added. The organic solvent was evaporated and the remaining aqueous phase was extracted with DCM. The combined organic extracts were washed with brine, dried over MgSO4, filtered and concentrated to dryness. The residue was purified by flash chromatography, silica gel, gradient DCM to MeOH:DCM (1:4), to give the title compound (0.48 g, 69% yield).
References:
WO2022/229405,2022,A1 Location in patent:Page/Page column 52

177906-48-8
213 suppliers
$6.00/250mg

264608-33-5
7 suppliers
inquiry