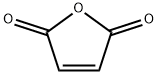
2-Butenoic acid, 4-[4-(1,1-dimethylethyl)phenyl]-4-oxo-, (2E)- synthesis
- Product Name:2-Butenoic acid, 4-[4-(1,1-dimethylethyl)phenyl]-4-oxo-, (2E)-
- CAS Number:29685-73-2
- Molecular formula:C14H16O3
- Molecular Weight:232.27
![2-Butenoic acid, 4-[4-(1,1-dimethylethyl)phenyl]-4-oxo-, ethyl ester, (2E)-](/CAS/20210305/GIF/1073375-56-0.gif)
1073375-56-0
0 suppliers
inquiry
![2-Butenoic acid, 4-[4-(1,1-dimethylethyl)phenyl]-4-oxo-, (2E)-](/CAS/20200401/GIF/29685-73-2.gif)
29685-73-2
3 suppliers
inquiry
Yield:29685-73-2 90%
Reaction Conditions:
with potassium carbonate in methanol;water at 60; for 5 h;Inert atmosphere;
Steps:
5.10 (E)-4-(4-(tert-Butyl)phenyl)-4-oxobut-2-enoic acid (16)
To the trans ester 13 (105mg, 0.4mmol) in CH3OH (5mL) was added K2CO3 (0.279mg, 2mmol) in H2O (5mL). The reaction mixture was allowed to reflux for 5h and then the CH3OH was removed under reduced pressure. The residue was then cooled to 0°C and brought to pH 2 with a solution of cold aq HCl (1M). The mixture, which resulted, was extracted with diethyl ether (2×15mL). The combined extracts were washed with brine (20mL), dried (Na2SO4) and concentrated under reduced pressure to furnish a pale green solid acid 16 (84.3mg, 90%). Mp 118-121°C. 1H NMR (300MHz, CDCl3): δ 8.03 (d, J=15.6Hz, 1H), 7.98 (d, J=7.97Hz, 2H), 7.56 (d, J=7.97Hz, 2H), 6.91 (d, J=15.3Hz, 2H), 1.38 (S, 9H); 13C NMR (75MHz, CDCl3): δ 188.8, 170.5, 158.2, 138.6, 133.8, 131.1, 128.9, 125.9, 35.3, 31.0. HRMS (ESI) (M+H)+, calcd for C14H17O3 233.1178; found 233.1155. This material was employed directly in the next experiment
References:
Tiruveedhula, V.V.N. Phani Babu;Witzigmann, Christopher M.;Verma, Ranjit;Kabir, M. Shahjahan;Rott, Marc;Schwan, William R.;Medina-Bielski, Sara;Lane, Michelle;Close, William;Polanowski, Rebecca L.;Sherman, David;Monte, Aaron;Deschamps, Jeffrey R.;Cook, James M. [Bioorganic and Medicinal Chemistry,2013,vol. 21,# 24,p. 7830 - 7840]
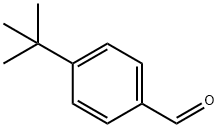
939-97-9
280 suppliers
$15.00/5g
![2-Butenoic acid, 4-[4-(1,1-dimethylethyl)phenyl]-4-oxo-, (2E)-](/CAS/20200401/GIF/29685-73-2.gif)
29685-73-2
3 suppliers
inquiry