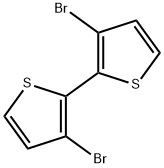
3,3'-Dibromo-2,2'-bithiophene synthesis
- Product Name:3,3'-Dibromo-2,2'-bithiophene
- CAS Number:51751-44-1
- Molecular formula:C8H4Br2S2
- Molecular Weight:324.06

The freshly prepared LDA solution (prepared by the addition ofn-BuLi (2.5 M in hexanes, 100 mmol, 40 mL) and diisopropylamine(11.1 g, 110 mmol) in 50 mL of anhydrous THF (78 C to roomtemperature)) was added drop wise to a solution of 3-bromothiophene (16.30 g, 100 mmol) in anhydrous THF (100 mL)at 78 C under nitrogen atmosphere. The reaction mixture wasstirred for 1 h at 78 C and CuCl2 (14.11 g, 105 mmol)was added inone portion. The reaction mixture was allowed towarm up to roomtemperature and treated with aqueous HCl. The organic phaseseparated was collected and the aqueous phase was extracted withdiethyl ether several times. The combined organic phase was driedover anhydrous Na2SO4 and the solution was filtered, concentratedand the residue was purified by silica gel column chromatography(hexane eluent) to afford a the required compound as white solid(yield 85%).1H NMR (500 MHz, CDCl3, d ppm): 7.08 (d, J 5.0 Hz, 2H), 7.40(d, J 5.0 Hz, 2H).
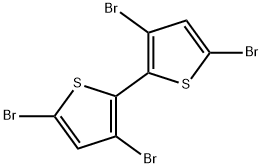
125143-53-5
129 suppliers
$5.00/250mg

51751-44-1
235 suppliers
$7.00/100mg
Yield:51751-44-1 92%
Reaction Conditions:
with hydrogenchloride;acetic acid;zinc in ethanol;water; for 2.5 h;Reflux;
Steps:
3 3,3′-dibromo-2,2′-bithiophene (11)
3,3′-dibromo-2,2′-bithiophene (11)
3,3′,5,5′-Tetrabromo-2,2′-bithiophene, 10 (15 g, 31.1 mmol) was added in portion within 0.5 h to a refluxing dispersion of zinc powder (7.8 g, 0.12 mol) in 150 mL of ethanol containing 15 mL of water, 72 mL of glacial acetic acid, and 3.1 mL of 3 M HCl. After refluxing for an additional 2 h and then cooling to room temperature, the mixture was filtered and washed three times with ethanol, and the filtrate was collected. The solvent was then removed by evaporation, and 60 mL of H2O was added. The mixture was then extracted with dichloromethane (3×100 mL), and the combined extract was washed with water, dried over anhydrous MgSO4, and filtered. The solvent was removed by evaporation, and the crude product was crystallized from hexane to give colorless crystals of product 9.2 g, 92% yield. 1H NMR (400 MHz, CDCl3, δ) 7.43 (d, J=5.4 Hz, 2H), 7.09 (d, J=5.4 Hz, 2H). 13C NMR (100 MHz, CDCl3, δ) 130.8, 128.8, 127.5, 112.6. MS (FAB): m/z 324.0 (M+).
References:
US8975419,2015,B2 Location in patent:Page/Page column 10; 11; 12; 17; 18
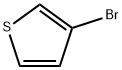
872-31-1
336 suppliers
$9.00/1g

51751-44-1
235 suppliers
$7.00/100mg
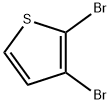
3140-93-0
294 suppliers
$6.00/5g

51751-44-1
235 suppliers
$7.00/100mg
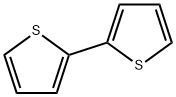
492-97-7
314 suppliers
$5.00/1g

51751-44-1
235 suppliers
$7.00/100mg
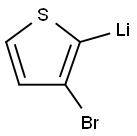
19162-82-4
0 suppliers
inquiry

51751-44-1
235 suppliers
$7.00/100mg