
3,4,4'-TRICHLOROBENZHYDROL synthesis
- Product Name:3,4,4'-TRICHLOROBENZHYDROL
- CAS Number:660840-68-6
- Molecular formula:C13H9Cl3O
- Molecular Weight:287.57
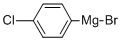
873-77-8
145 suppliers
$61.21/100ml
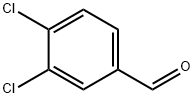
6287-38-3
252 suppliers
$8.00/10g

660840-68-6
21 suppliers
$809.00/1g
Yield:-
Reaction Conditions:
Stage #1: (4-chlorphenyl)magnesium bromide;3,4-dichlorobenzaldehyde in diethyl ether at 0 - 20; for 4.5 h;Heating / reflux;
Stage #2: with hydrogenchloride in diethyl ether;water;
Steps:
30.A Step A: [(4-Chlorophenyl)-(3,4-dichlorophenyl)methanol
To a solution of 3,4-dichlorobenzaldehyde (5.3 g, 30 mmol) in dry Ether (50 mL) under nitrogen atmosphere cooled in an ice-bath was added a 1 M solution of 4-chlorophenylmagnesium bromide in Ether (33 [ML,] 33 mmol) dropwise. The resulting mixture was stirred 0.5 hours, warmed to room temperature, stirred 2 hours, warmed to reflux, and refluxed 2 hours. After cooling to room temperature, the reaction mixture was quenched by the careful addition of 2N [HC1,] diluted with ether (100 mL) and washed with brine. The organic phase was dried over [MGSO4] and concentrated under reduced pressure. The residue was purified by flash chromatography (EtOAc/Hexane, 1: 9) to give 5.3 g of a white solid. 1H NMR CDC13 8 2.50 (d, J = 3.3 Hz), 5.76 (d, J = 3.3 Hz), 7.15-7. 47 (m, 7H). MS 286 (M-1). Anal. [(CL3H9CL3O)] C, H, N.
References:
WO2004/14847,2004,A1 Location in patent:Page 94-95