
3,6-DIBROMO-9-(OXIRAN-2-YLMETHYL)-9H-CARBAZOLE synthesis
- Product Name:3,6-DIBROMO-9-(OXIRAN-2-YLMETHYL)-9H-CARBAZOLE
- CAS Number:85446-05-5
- Molecular formula:C15H11Br2NO
- Molecular Weight:381.06
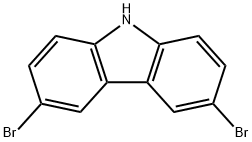
6825-20-3
326 suppliers
$5.00/5g

3132-64-7
119 suppliers
$20.00/1g

85446-05-5
28 suppliers
$28.60/25MG
Yield:85446-05-5 85.7%
Reaction Conditions:
Stage #1: 3,6-dibromo-9H-carbazolewith potassium hydroxide in N,N-dimethyl-formamide at 20; for 1 h;
Stage #2: 1,2-Epoxy-3-bromopropane in N,N-dimethyl-formamide at 20; for 3 h;
Steps:
1.a
a. Dissolve 6g 3,6-dibromocarbazole in 60mL N,N-dimethylformamide,Add 1.2g of potassium hydroxide powder, stir at room temperature for 1h,6.2g epibromohydrin was added dropwise, and reacted at room temperature for 3 hours.The reaction solution was diluted with 100 mL of water, filtered, and the solid was collected.Drying to obtain a white solid compound 1 (6 g, 85.7%).
References:
CN113402444,2021,A Location in patent:Paragraph 0072-0075

6825-20-3
326 suppliers
$5.00/5g
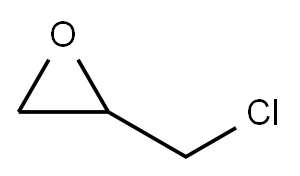
106-89-8
734 suppliers
$9.00/10g

85446-05-5
28 suppliers
$28.60/25MG

6825-20-3
326 suppliers
$5.00/5g

556-52-5
251 suppliers
$17.67/1gm:

85446-05-5
28 suppliers
$28.60/25MG
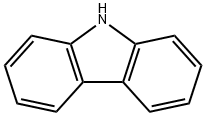
86-74-8
323 suppliers
$10.00/5g

85446-05-5
28 suppliers
$28.60/25MG